For most of chemistry, atoms are treated like indestructible pieces that simply rearrange. Oxygen atoms stay oxygen, carbon atoms stay carbon, and chemical equations balance because the same atoms appear before and after the reaction. Nuclear science breaks that expectation. In a nuclear process, an atom of one element can turn into an atom of a different element. That may sound almost impossible at first, but it is one of the deepest discoveries in modern science.
The key idea is this: atoms are not conserved in nuclear processes, because the nucleus can change. However, the total number of particles in the nucleus, meaning protons plus neutrons, is conserved in the reaction equation. This is why nuclear equations are balanced differently from ordinary chemical equations. To understand that, we first need to look closely at what is inside an atom.
[Figure 1] At the center of every atom is the nucleus, a tiny, dense region containing protons and neutrons. Protons have positive charge, neutrons have no charge, and together they make up almost all of the atom's mass. The number of protons determines the element. A nucleus with \(6\) protons is carbon; a nucleus with \(8\) protons is oxygen. Counting protons identifies the element, while counting protons and neutrons together identifies the specific nuclide.
The atomic number is the number of protons. The mass number is the total number of protons plus neutrons. For example, \(\,^{14}_{6}C\,\) is carbon-14. The lower number, \(6\), is the atomic number, so this nucleus has \(6\) protons. The upper number, \(14\), is the mass number, so it has \(14 - 6 = 8\) neutrons.
Atoms of the same element can have different numbers of neutrons. These are called isotopes. Carbon-12 and carbon-14 are both carbon because both have \(6\) protons, but they are different isotopes because they have different numbers of neutrons. Isotopes often behave nearly the same in chemical reactions because chemistry depends mostly on electrons, but they can behave very differently in nuclear processes.
Atomic number is the number of protons in a nucleus.
Mass number is the total number of protons plus neutrons in a nucleus.
Isotopes are atoms of the same element that have the same number of protons but different numbers of neutrons.
This distinction matters because nuclear changes happen in the nucleus, not in the electron cloud. If the number of protons changes, the atom becomes a different element. That is why nuclear reactions can transform matter in a way that chemical reactions cannot.
In an ordinary chemical reaction such as \[2H_2 + O_2 \rightarrow 2H_2O\], the hydrogen atoms and oxygen atoms are conserved. They are rearranged into new molecules, but hydrogen stays hydrogen and oxygen stays oxygen. No nuclei are changed.
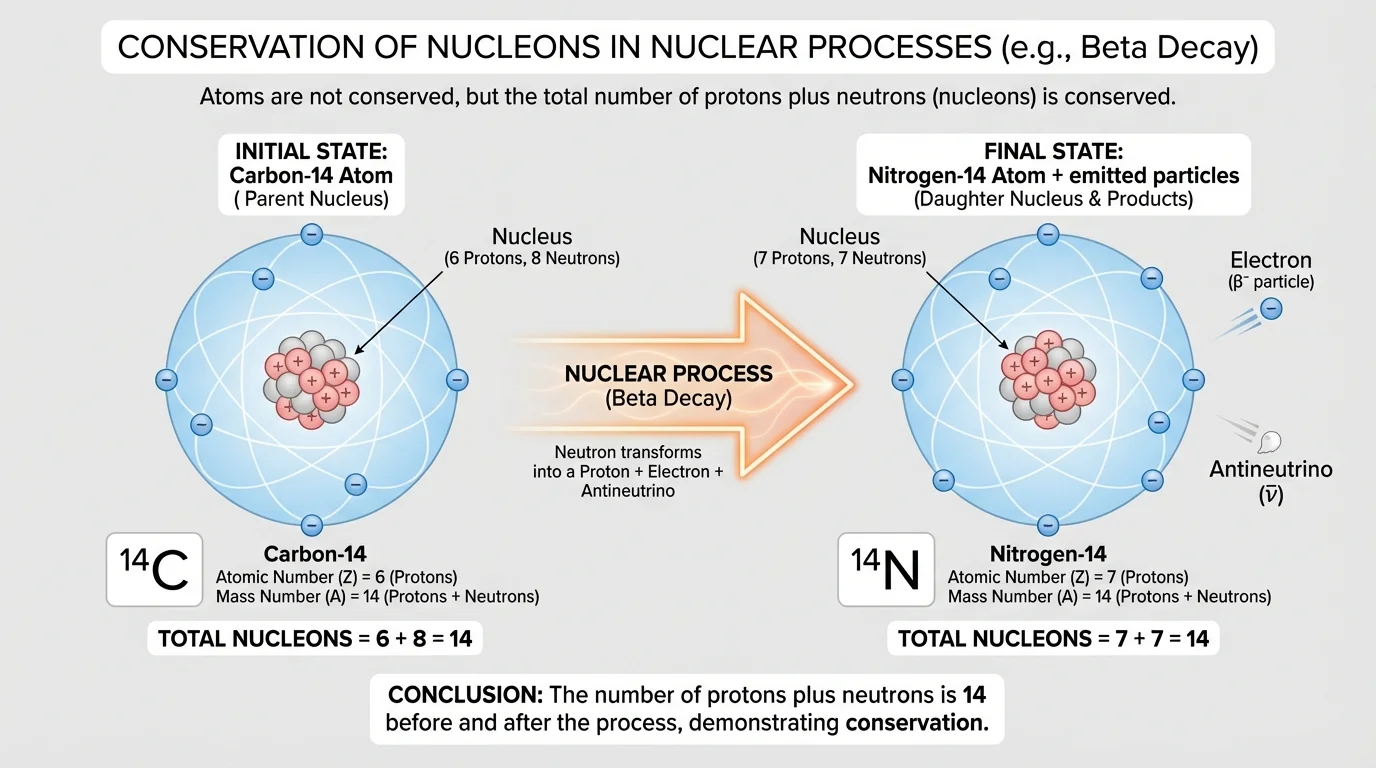
In a nuclear reaction, the nucleus itself changes. A neutron can turn into a proton, a heavy nucleus can split into smaller nuclei, or two light nuclei can combine. Because the number of protons can change, the identity of the atom can change. For example, carbon can become nitrogen in a nuclear process. That would never happen in a normal chemical reaction.
So when scientists say atoms are not conserved in nuclear processes, they mean the number of each kind of atom is not fixed. The reaction may begin with one element and end with another. What must still balance in the nuclear equation is the total number of nucleons, meaning protons plus neutrons, along with electric charge.
Why chemistry and nuclear science balance equations differently
Chemical equations conserve each type of atom because nuclei remain unchanged. Nuclear equations conserve total nucleons and charge because nuclei are rearranged internally. The element symbols on the two sides do not have to match, because one nucleus can transform into another.
This is one reason nuclear science was so revolutionary. Ancient thinkers imagined one substance changing into another, but modern nuclear physics showed that element transformation is real under specific conditions inside the nucleus.
To balance a nuclear equation, track two numbers for every nucleus: atomic number and mass number. The sum of the atomic numbers must be the same on both sides, and the sum of the mass numbers must also be the same on both sides.
For example, consider alpha decay of uranium-238:
\[\,^{238}_{92}U \rightarrow \,^{234}_{90}Th + \,^{4}_{2}He\]
Check the balance. For mass number, \(238 = 234 + 4\). For atomic number, \(92 = 90 + 2\). The atom is not conserved because uranium becomes thorium, but the total number of protons plus neutrons is conserved.
Balancing a nuclear equation
Find the missing particle in \[\,^{14}_{6}C \rightarrow \,^{14}_{7}N + ?\]
Step 1: Compare mass numbers.
On the left, the mass number is \(14\). On the right, nitrogen already has mass number \(14\), so the missing particle must have mass number \(0\).
Step 2: Compare atomic numbers.
On the left, the atomic number is \(6\). On the right, nitrogen has atomic number \(7\), so the missing particle must contribute \(-1\) to balance the equation.
Step 3: Identify the particle.
A particle with mass number \(0\) and atomic number \(-1\) is a beta particle, written as \(\,^{0}_{-1}e\,\).
The balanced equation is \[\,^{14}_{6}C \rightarrow \,^{14}_{7}N + \,^{0}_{-1}e\]
Notice something subtle here: the total number of nucleons stays \(14\), but one neutron inside the carbon nucleus has changed into a proton. That raises the atomic number from \(6\) to \(7\), changing carbon into nitrogen.
[Figure 2] Some nuclei are unstable and change on their own. This spontaneous process is called radioactive decay. The three common types discussed in high school are alpha decay, beta decay, and gamma emission. Each changes the nucleus in a different way, especially in how proton and neutron counts are affected.
Alpha decay happens when a heavy nucleus emits an alpha particle, which is a helium nucleus \(\,^{4}_{2}He\,\). This lowers the mass number by \(4\) and the atomic number by \(2\). Heavy elements such as uranium and radium commonly undergo alpha decay.
Beta decay usually means beta minus decay in introductory courses. In this process a neutron changes into a proton and an electron is emitted. The mass number stays the same, but the atomic number increases by \(1\). This is why \(\,^{14}_{6}C\,\) becomes \(\,^{14}_{7}N\,\).
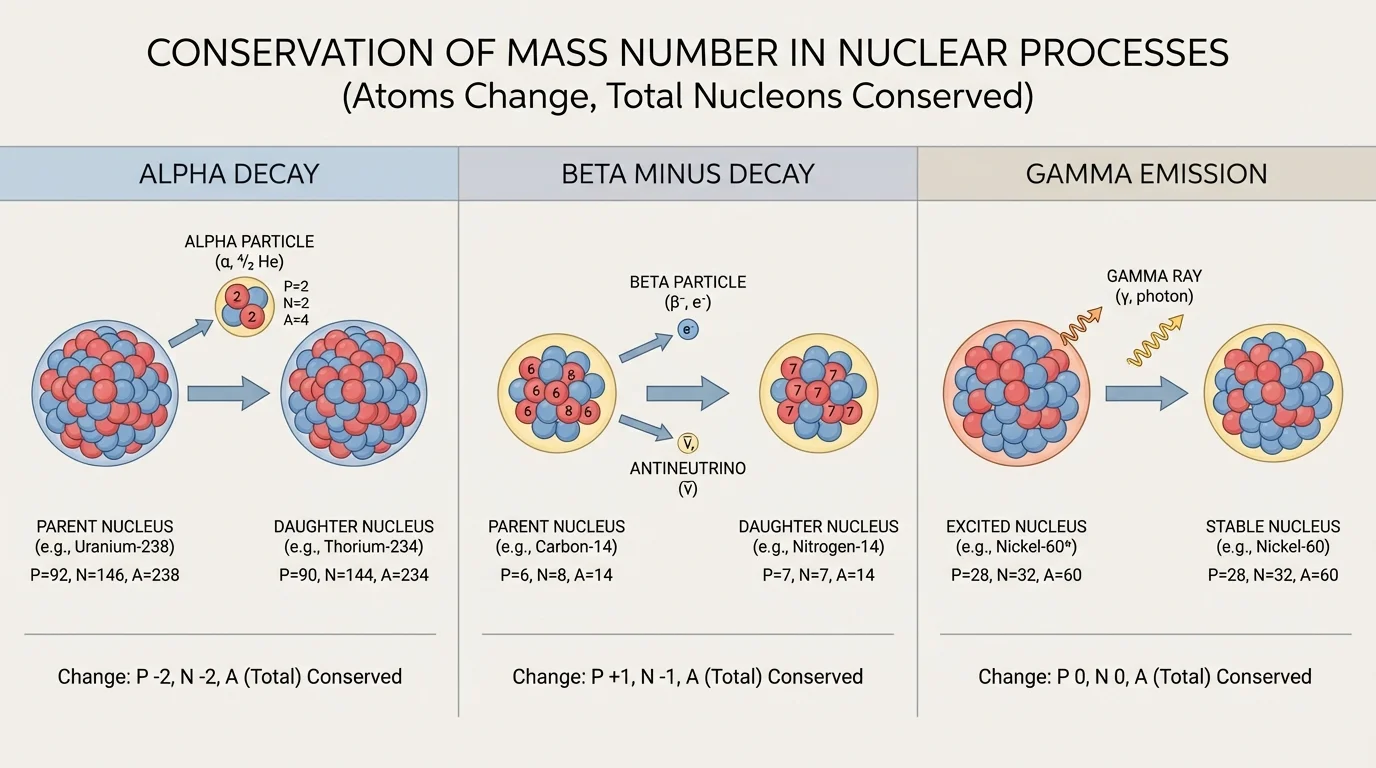
Gamma emission is different. The nucleus releases energy as gamma radiation, but the numbers of protons and neutrons do not change. The nucleus goes from a higher-energy state to a lower-energy state. So the atom stays the same element and the same isotope, but it loses nuclear energy.
A useful way to remember this is to ask two questions: Does the nucleus lose or gain nucleons, and does the number of protons change? Alpha decay changes both. Beta decay changes the proton count but not the total nucleon count. Gamma emission changes neither count; it only lowers energy.
Carbon-14 dating works because carbon-14 is radioactive and slowly decays into nitrogen-14. By measuring how much carbon-14 remains in once-living material, scientists can estimate its age.
Later, when you compare nuclear power and medical tracers, this same decay logic matters. The pattern in [Figure 2] helps explain why different isotopes have different uses and risks depending on the type and energy of the radiation they emit.
[Figure 3] Fission is the splitting of a heavy nucleus into smaller nuclei. A famous example involves uranium-235. When \(\,^{235}_{92}U\,\) absorbs a neutron, it can become unstable and split into two medium-sized nuclei plus more neutrons. One large nucleus breaks apart and releases additional neutrons that can trigger more fission events.
One possible fission reaction is:
\[\,^{235}_{92}U + \,^{1}_{0}n \rightarrow \,^{141}_{56}Ba + \,^{92}_{36}Kr + 3\,^{1}_{0}n\]
Check the totals. The mass numbers give \(235 + 1 = 141 + 92 + 3\), so \(236 = 236\). The atomic numbers give \(92 = 56 + 36\). The original uranium atom is not conserved, because it becomes barium and krypton, but the total nucleons are conserved.
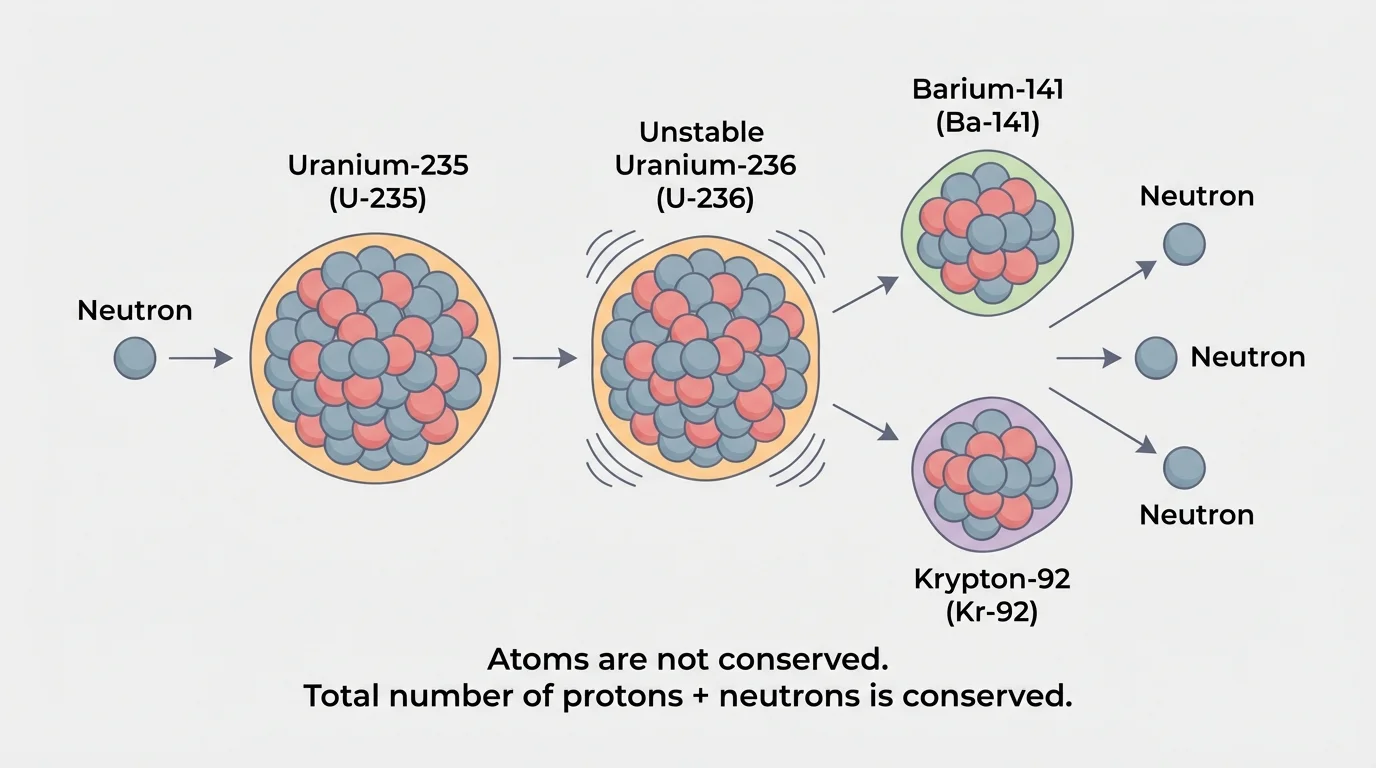
The emitted neutrons are extremely important. If they strike other uranium-235 nuclei, they can cause additional fission reactions. This creates a chain reaction. In a nuclear reactor, the chain reaction is controlled so energy is released steadily. In a nuclear weapon, the chain reaction occurs extremely rapidly.
Fission releases a huge amount of energy compared with chemical reactions. Burning coal or gasoline rearranges electrons in chemical bonds. Fission reorganizes the nucleus, where the energy scale is far larger. That is why a small amount of nuclear fuel can produce so much electricity.
Checking conservation in fission
Verify that \[\,^{235}_{92}U + \,^{1}_{0}n \rightarrow \,^{141}_{56}Ba + \,^{92}_{36}Kr + 3\,^{1}_{0}n\] is balanced.
Step 1: Add the mass numbers on each side.
Left side: \(235 + 1 = 236\). Right side: \(141 + 92 + 3 = 236\).
Step 2: Add the atomic numbers on each side.
Left side: \(92 + 0 = 92\). Right side: \(56 + 36 + 0 = 92\).
Step 3: Interpret the result.
The equation is balanced, so both total nucleons and charge are conserved even though uranium turns into different elements.
Reactor design depends on this balance. Engineers track how many neutrons are produced, absorbed, or lost, because controlling neutron flow controls the chain reaction shown earlier in [Figure 3].
[Figure 4] Fusion is the joining of light nuclei to form a heavier nucleus. Fusion powers the Sun and other stars, although stars mainly fuse hydrogen through multistep pathways. One important fusion reaction studied for energy technology is the combination of deuterium and tritium, which are isotopes of hydrogen, to form helium and a neutron. Two small nuclei combine into a more tightly bound nucleus while releasing energy.
A common fusion equation is:
\[\,^{2}_{1}H + \,^{3}_{1}H \rightarrow \,^{4}_{2}He + \,^{1}_{0}n\]
Again, check conservation. Mass numbers: \(2 + 3 = 4 + 1\), so \(5 = 5\). Atomic numbers: \(1 + 1 = 2 + 0\), so \(2 = 2\). The original hydrogen atoms are not conserved as hydrogen atoms, because they become helium, but the total number of protons plus neutrons is conserved.
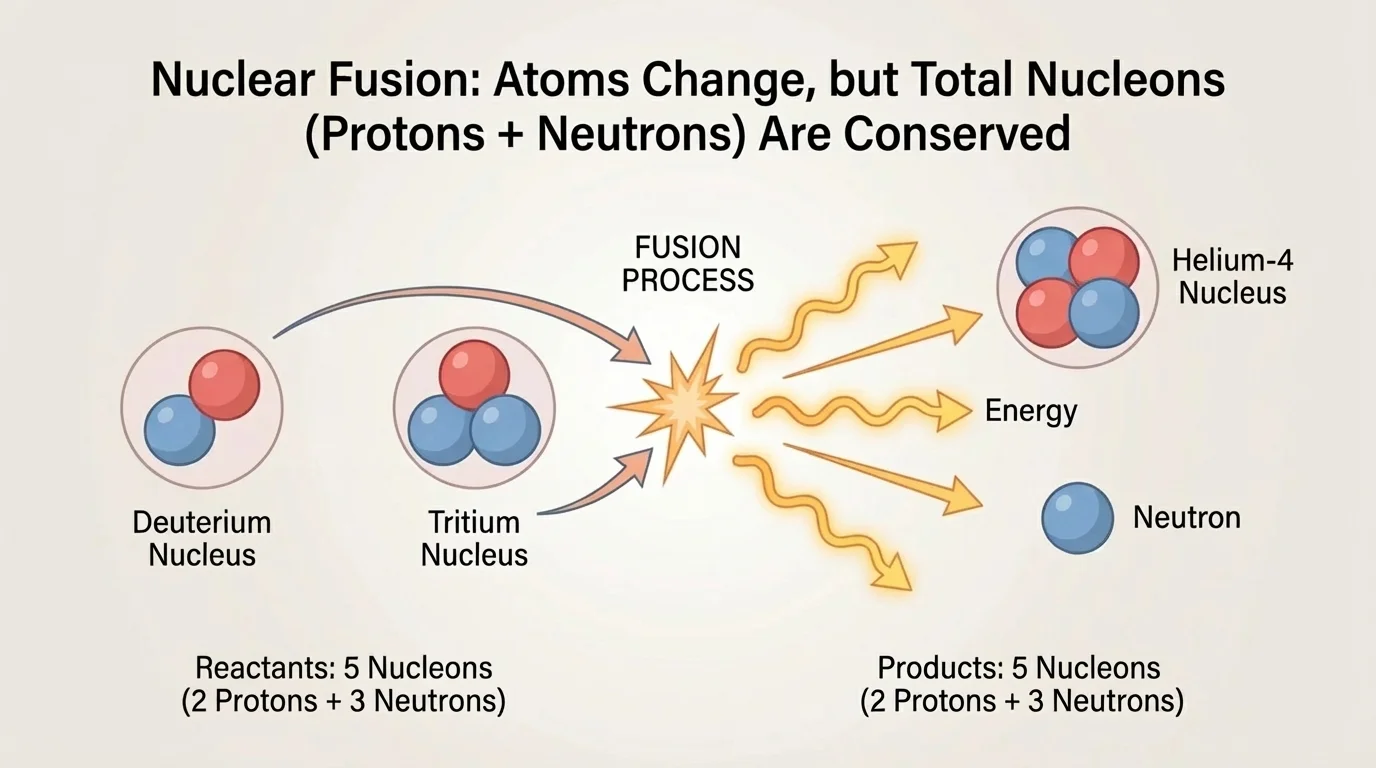
Fusion is difficult on Earth because positively charged nuclei repel each other. To overcome this repulsion, nuclei must move extremely fast, which requires very high temperature. In stars, immense pressure and temperature make fusion possible. On Earth, scientists are trying to achieve controlled fusion for energy production, but it remains technologically challenging.
Fusion is attractive as an energy source because light fuels can release enormous energy and produce less long-lived radioactive waste than fission. Still, the temperatures involved are so high that containing the reacting plasma is a major engineering problem.
Chemical bond energy comes from changes in electron arrangements outside the nucleus. Nuclear energy comes from changes inside the nucleus itself, so it is typically millions of times larger per atom.
The comparison between fission and fusion becomes clearer if you look back at [Figure 4]. In both cases, the products are more stable in nuclear terms than the starting nuclei, and that stability difference is connected to the released energy.
A natural question follows: if the total number of protons plus neutrons is conserved, where does the released energy come from? The answer involves mass defect and binding energy. The mass of a bound nucleus is slightly less than the total mass of its separate protons and neutrons. That missing mass corresponds to energy.
Einstein's relation connects mass and energy:
\(E = mc^2\)
Here, \(E\) is energy, \(m\) is mass, and \(c\) is the speed of light. Because \(c^2\) is enormous, even a tiny mass change produces a large amount of energy.
Mass-energy example
Suppose a nuclear reaction converts \(1.0 \times 10^{-6}\,\mathrm{kg}\) of mass into energy. Find the energy released using \(E = mc^2\), with \(c = 3.0 \times 10^8\,\mathrm{m/s}\).
Step 1: Square the speed of light.
\(c^2 = (3.0 \times 10^8)^2 = 9.0 \times 10^{16}\)
Step 2: Multiply by the mass.
\(E = (1.0 \times 10^{-6})(9.0 \times 10^{16}) = 9.0 \times 10^{10}\,\mathrm{J}\)
Step 3: Interpret the result.
This is an enormous amount of energy from a very small mass change.
\[E = 9.0 \times 10^{10}\,\mathrm{J}\]
This does not mean nucleons disappear randomly. It means the total mass of the final bound system is slightly lower than the initial system, and the difference emerges as energy. In fission and fusion, the products are more tightly bound, so some mass is released as kinetic energy and radiation.
The contrast between chemical and nuclear processes is easier to see side by side.
| Feature | Chemical reaction | Nuclear process |
|---|---|---|
| What changes? | Electron arrangement and bonding | Nucleus composition |
| Are atoms of each element conserved? | Yes | No |
| Is total number of protons plus neutrons conserved? | Usually unchanged because nuclei do not change | Yes, in balanced nuclear equations |
| Can one element become another? | No | Yes |
| Typical energy scale | Relatively small | Very large |
Table 1. Comparison of chemical reactions and nuclear processes.
This is why a statement that sounds contradictory is actually precise: atoms are not conserved in nuclear processes, but nucleons are conserved. The nucleus can be rearranged into a different element while still following strict conservation rules.
Nuclear ideas are not just abstract physics. They affect energy systems, medicine, archaeology, environmental science, and national security. Nuclear power plants use controlled fission to heat water, drive turbines, and generate electricity. Medical imaging uses radioactive isotopes that emit detectable radiation. Radiation therapy targets cancer cells by damaging their DNA.
Smoke detectors often use a tiny amount of americium-241, whose radiation helps detect particles from smoke. Carbon dating uses radioactive decay to estimate the age of biological remains. Industrial radiography uses radiation to inspect metal parts and welds without cutting them open.
Why shielding depends on the type of radiation
Alpha particles are blocked easily, even by paper or skin, but are dangerous if alpha-emitting material is inhaled or swallowed. Beta particles penetrate farther and may require plastic or metal shielding. Gamma rays are highly penetrating and often require dense shielding such as lead or thick concrete.
These benefits come with serious risks. Uncontrolled chain reactions, radiation exposure, long-term waste storage, and the possibility of weapons use make nuclear technology ethically and politically important. Scientific understanding is essential because fear without knowledge can be misleading, but confidence without caution can be dangerous.
One misconception is that conservation of nucleon number means the mass itself never changes. In fact, measurable mass can decrease slightly because some mass is converted to energy. Another misconception is that gamma decay changes the element. It does not; it only changes the energy state of the nucleus.
A third misconception is that nuclear equations balance by counting the same atoms on both sides. They do not. A uranium nucleus may become thorium, or hydrogen may become helium. What balances is the total mass number and total atomic number.
Finally, students sometimes confuse "protons plus neutrons are conserved" with "every single proton and neutron stays the same particle forever." In some processes, such as beta decay, a neutron can transform into a proton. What remains conserved in the equation is the total number of nucleons, not necessarily the separate counts of protons and neutrons.
"The nucleus is small, but the changes within it can transform elements and release extraordinary energy."
That is the heart of nuclear science: the atom is not an unchangeable object. Under nuclear processes, the nucleus can change composition, the element can change identity, and energy can be released on a remarkable scale, all while the total number of protons plus neutrons remains conserved in the balanced nuclear description.