Your body is made partly from yesterday's lunch and partly from air. That sounds strange, but it is true. The carbon in your muscles may once have been in a plant leaf, then in a sandwich, and later in your body. The amazing part is that this matter does not vanish or magically appear. It gets rearranged. That is the big idea of this lesson: matter is conserved because atoms are conserved.
Every time you eat, breathe, grow, run, or even sit still, your body is carrying out physical and chemical processes. In these processes, substances change form, but the total amount of matter stays the same. This idea helps explain how food becomes body tissue, how plants make sugars, and why scientists use balanced equations to describe reactions.
If matter could just disappear, science would be much harder to understand. But nature follows patterns. Atoms move, join, separate, and rearrange in predictable ways. That is why scientists can trace matter through living things and through the environment.
You already know that matter has mass and takes up space. You also know that substances can change state, such as solid ice melting into liquid water. This lesson builds on that idea by showing that even when substances change a lot, the atoms are still there.
To understand conservation of matter, we need to look closely at what matter is made of.
Atoms are tiny particles that make up all matter. A molecule is a group of atoms joined together. The key idea, as shown in [Figure 1], is that atoms can be separated from one group and joined into another group, but the atoms themselves are still present before and after the change.
For example, water is made of molecules with the formula \(\textrm{H}_2\textrm{O}\). Each water molecule contains \(2\) hydrogen atoms and \(1\) oxygen atom. If water freezes into ice, those same atoms are still there. If water evaporates into water vapor, those same atoms are still there. The arrangement and movement of particles change, but the atoms are conserved.
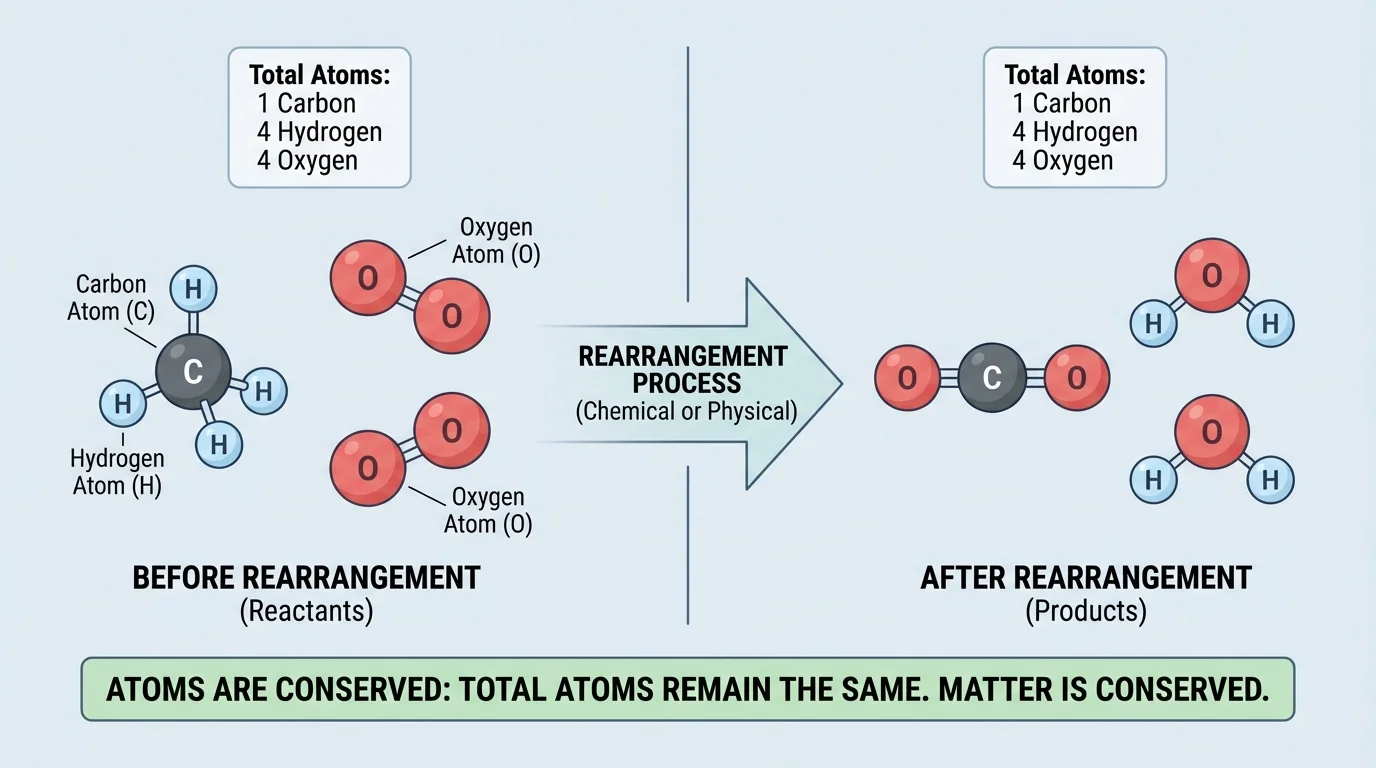
Atoms combine in many ways. Carbon, hydrogen, and oxygen can join to form sugar molecules such as \(\textrm{C}_6\textrm{H}_{12}\textrm{O}_6\). They can also be part of fats, proteins, and many other molecules in living things. Even though molecules may be large and complex, they still follow the same rule: atoms are rearranged, not destroyed.
Matter is anything that has mass and takes up space.
Conservation of matter means the total amount of matter stays the same during physical and chemical processes.
A chemical reaction is a process in which atoms are rearranged to form new substances.
This is why scientists often think about reactions as a kind of atom bookkeeping. They count the atoms on one side and make sure the same number of each kind appears on the other side.
A physical change changes the form, size, or state of a substance, but it does not create a new substance. Melting ice, tearing paper, crushing a can, and dissolving sugar in water are common examples.
Suppose a piece of ice melts. Before melting, the particles are arranged in a solid pattern. After melting, the particles move more freely as a liquid. But the substance is still water, \(\textrm{H}_2\textrm{O}\). The number of hydrogen atoms and oxygen atoms stays exactly the same.
Dissolving can be trickier. When table salt, \(\textrm{NaCl}\), dissolves in water, it may seem to disappear. It has not. The particles have spread out in the water, but the sodium and chlorine atoms are still present. If the water evaporates, the salt can often be collected again.
In physical changes, matter may look different, but no atoms are lost. They are simply spaced differently, moving differently, or mixed differently.
A chemical reaction forms new substances. The products are different from the reactants, but conservation still holds. Balanced equations, as introduced in [Figure 2], help us show that the number of each kind of atom remains equal before and after the reaction.
Consider the formation of water from hydrogen gas and oxygen gas:
\[2\textrm{H}_2 + \textrm{O}_2 \rightarrow 2\textrm{H}_2\textrm{O}\]
On the left side, there are \(4\) hydrogen atoms and \(2\) oxygen atoms. On the right side, there are also \(4\) hydrogen atoms and \(2\) oxygen atoms. The atoms have been rearranged into water molecules, but none have disappeared.
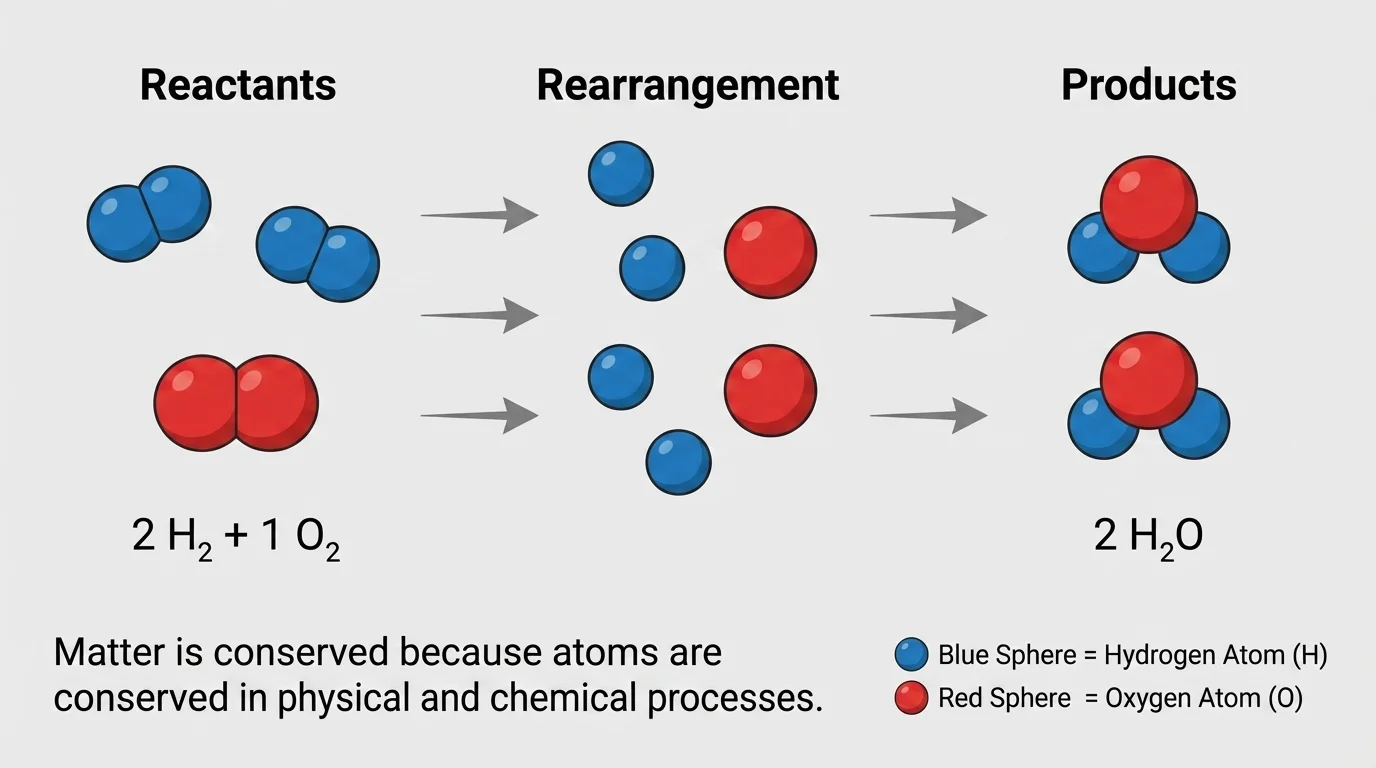
This is why equations must be balanced. An equation that is not balanced would suggest atoms were created or destroyed, which does not happen in ordinary physical and chemical processes.
Here is another example, the reaction used by plants and animals during life processes:
\[\textrm{C}_6\textrm{H}_{12}\textrm{O}_6 + 6\textrm{O}_2 \rightarrow 6\textrm{CO}_2 + 6\textrm{H}_2\textrm{O}\]
If you count carefully, both sides contain \(6\) carbon atoms, \(12\) hydrogen atoms, and \(18\) oxygen atoms. The molecules change, but the atoms are conserved.
Counting atoms in a reaction
Check whether the equation \(2\textrm{H}_2 + \textrm{O}_2 \rightarrow 2\textrm{H}_2\textrm{O}\) shows conservation of matter.
Step 1: Count atoms in the reactants.
\(2\textrm{H}_2\) means \(2 \times 2 = 4\) hydrogen atoms. \(\textrm{O}_2\) means \(2\) oxygen atoms.
Step 2: Count atoms in the products.
\(2\textrm{H}_2\textrm{O}\) means \(2\) water molecules. Together they contain \(4\) hydrogen atoms and \(2\) oxygen atoms.
Step 3: Compare both sides.
The numbers match: hydrogen \(4 = 4\), oxygen \(2 = 2\).
The equation is balanced, so it correctly shows that atoms are conserved.
When you see a balanced chemical equation, think of it as evidence that matter has not been lost. The same idea will help us understand what happens to food in living organisms.
As shown in [Figure 3], food is not just "fuel." It is also raw material for the body. Matter from food moves through an organism, and its atoms are rearranged through digestion, growth, repair, and energy-releasing reactions.
When you eat carbohydrates, fats, and proteins, your digestive system breaks large food molecules into smaller molecules. For example, starch can be broken into simple sugars, and proteins can be broken into amino acids. These smaller molecules can move into the bloodstream and then into cells.
Inside cells, the molecules may be used in different ways. Some are rearranged to build new body molecules. That is how your body grows hair, muscle, skin, and bone. Other molecules are broken down in reactions that release energy the cell can use.
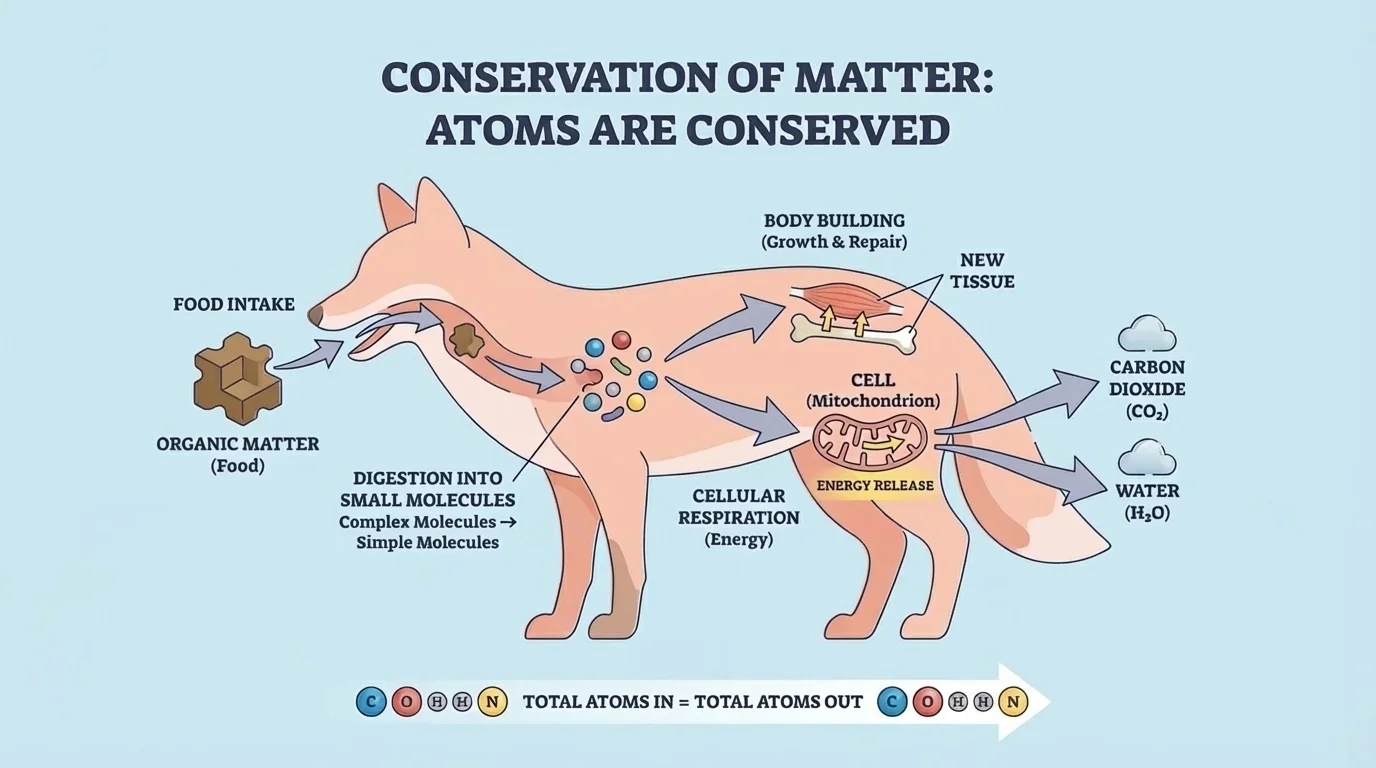
Notice something important: growth does not mean matter appears from nowhere. If a student gains \(2\) kilograms of body mass over time, that added matter came from food and water, with some atoms also coming from the oxygen breathed in. Body mass is built from matter entering the organism.
One major energy-releasing reaction in cells is cellular respiration. In simplified form, it can be written as:
\[\textrm{C}_6\textrm{H}_{12}\textrm{O}_6 + 6\textrm{O}_2 \rightarrow 6\textrm{CO}_2 + 6\textrm{H}_2\textrm{O} + \textrm{energy}\]
Cells use glucose, \(\textrm{C}_6\textrm{H}_{12}\textrm{O}_6\), and oxygen, \(\textrm{O}_2\), to produce carbon dioxide, \(\textrm{CO}_2\), and water, \(\textrm{H}_2\textrm{O}\), while releasing energy. The energy is not matter, but the atoms in the reactants become the atoms in the products.
How food supports growth and releases energy
Food molecules can follow more than one path inside an organism. Some are rearranged into body structures such as cell membranes, enzymes, and muscle proteins. Others are broken apart in cellular respiration. In both cases, atoms are conserved. The same carbon atom that was once in a sugar molecule may later become part of a muscle protein or may leave the body as carbon dioxide.
This means that when you exhale, some of the matter leaving your body contains atoms that were once part of your food. Later, in plants, those atoms may be used again.
As shown in [Figure 4], plants do something remarkable. They build sugars from carbon dioxide in the air and water from the soil. This process is called photosynthesis.
A simplified photosynthesis equation is:
\[6\textrm{CO}_2 + 6\textrm{H}_2\textrm{O} \rightarrow \textrm{C}_6\textrm{H}_{12}\textrm{O}_6 + 6\textrm{O}_2\]
Light energy helps drive this reaction. Notice the atom counts. On the left are \(6\) carbon atoms, \(12\) hydrogen atoms, and \(18\) oxygen atoms. On the right, the glucose and oxygen together still contain \(6\) carbon atoms, \(12\) hydrogen atoms, and \(18\) oxygen atoms.
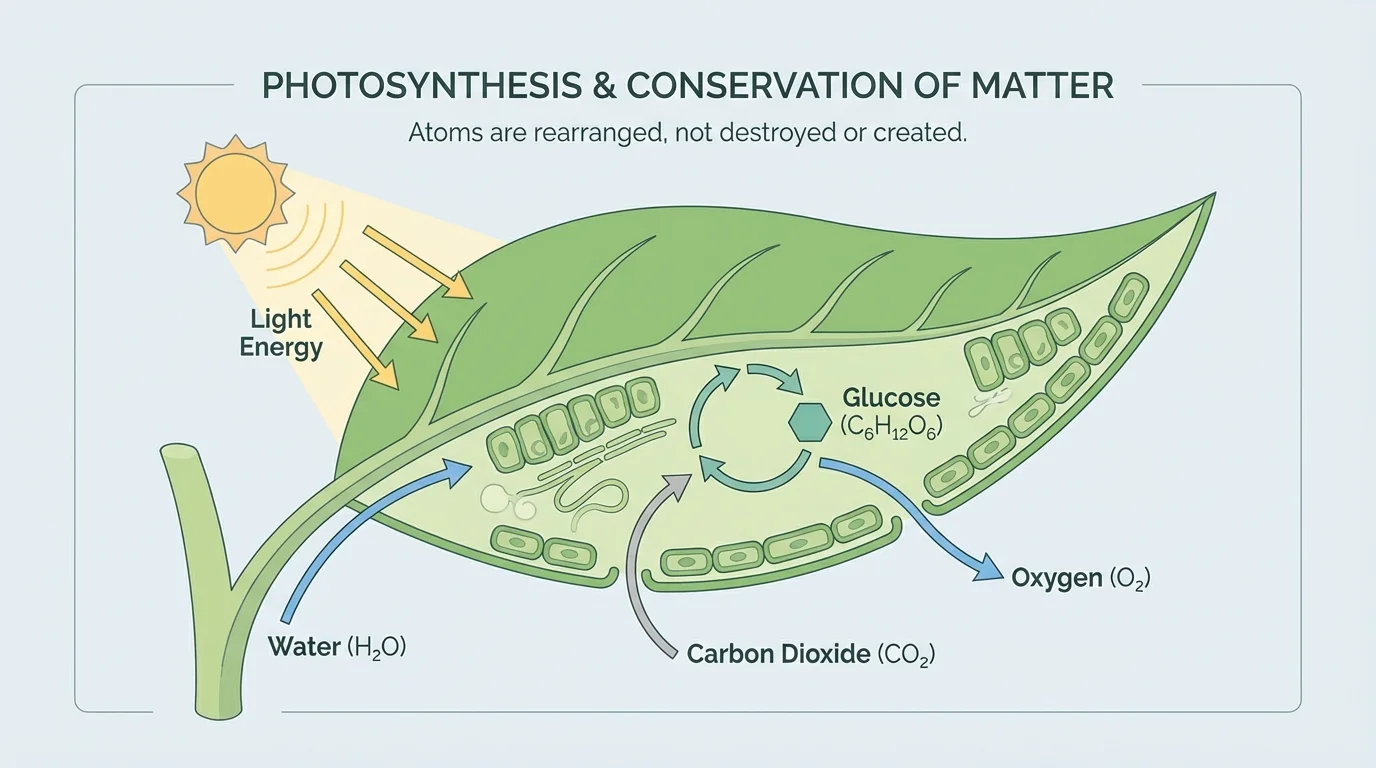
Photosynthesis explains how plant mass increases. A tree does not grow mainly by taking solid matter from the ground and turning it into wood. Much of its mass comes from carbon dioxide in the air. The carbon atoms become part of sugars, cellulose, and other plant molecules.
Later, animals may eat the plant. Then those same atoms move into the animal's body. This is one way matter moves through ecosystems.
Much of the solid material in a large tree comes from carbon dioxide gas in the air. Gas seems almost weightless, but over time its atoms can be built into trunks, branches, and roots.
The relationship between photosynthesis and cellular respiration is especially important. Plants use photosynthesis to build glucose. Animals and plants use cellular respiration to break down glucose and release energy. These processes recycle matter in living systems.
Let's trace a carbon atom. A plant takes in \(\textrm{CO}_2\) from the air. Through photosynthesis, that carbon atom may become part of a glucose molecule. If a rabbit eats the plant, digestion breaks the food into smaller molecules. The rabbit may use that carbon atom to build body tissue, or it may release it later as \(\textrm{CO}_2\) during cellular respiration.
The same type of tracing can be done for hydrogen and oxygen atoms. Hydrogen atoms from water may become part of sugars in plants or part of body molecules in animals. Oxygen atoms may be found in water, carbon dioxide, sugars, or many other compounds.
As we saw earlier, matter entering an organism can leave again in different forms. Food becomes body matter, waste, carbon dioxide, and water. Conservation of matter helps us connect all these changes into one story.
| Process | Matter going in | Matter coming out | What happens to atoms? |
|---|---|---|---|
| Melting ice | \(\textrm{H}_2\textrm{O}\) solid | \(\textrm{H}_2\textrm{O}\) liquid | Same atoms, different state |
| Dissolving salt | \(\textrm{NaCl}\) and \(\textrm{H}_2\textrm{O}\) | Sodium and chloride ions mixed in water | Same atoms, spread out |
| Cellular respiration | \(\textrm{C}_6\textrm{H}_{12}\textrm{O}_6\) and \(\textrm{O}_2\) | \(\textrm{CO}_2\) and \(\textrm{H}_2\textrm{O}\) | Atoms rearranged into new molecules |
| Photosynthesis | \(\textrm{CO}_2\) and \(\textrm{H}_2\textrm{O}\) | \(\textrm{C}_6\textrm{H}_{12}\textrm{O}_6\) and \(\textrm{O}_2\) | Atoms rearranged into new molecules |
Table 1. Comparison of several physical and chemical processes showing that atoms are conserved.
This way of tracking atoms is powerful because it shows that living things do not break the rules of chemistry. Life depends on rearranging matter, not creating it from nothing.
[Figure 5] helps explain why matter sometimes seems to disappear. This usually happens because some products are gases that spread into the air, making them harder to see or measure in an open system.
For example, when wood burns, ash is left behind, and students may think some matter was destroyed. But most of the matter became gases such as \(\textrm{CO}_2\) and water vapor that moved into the air. If all the products could be collected and measured in a closed system, the total mass would match the starting mass.
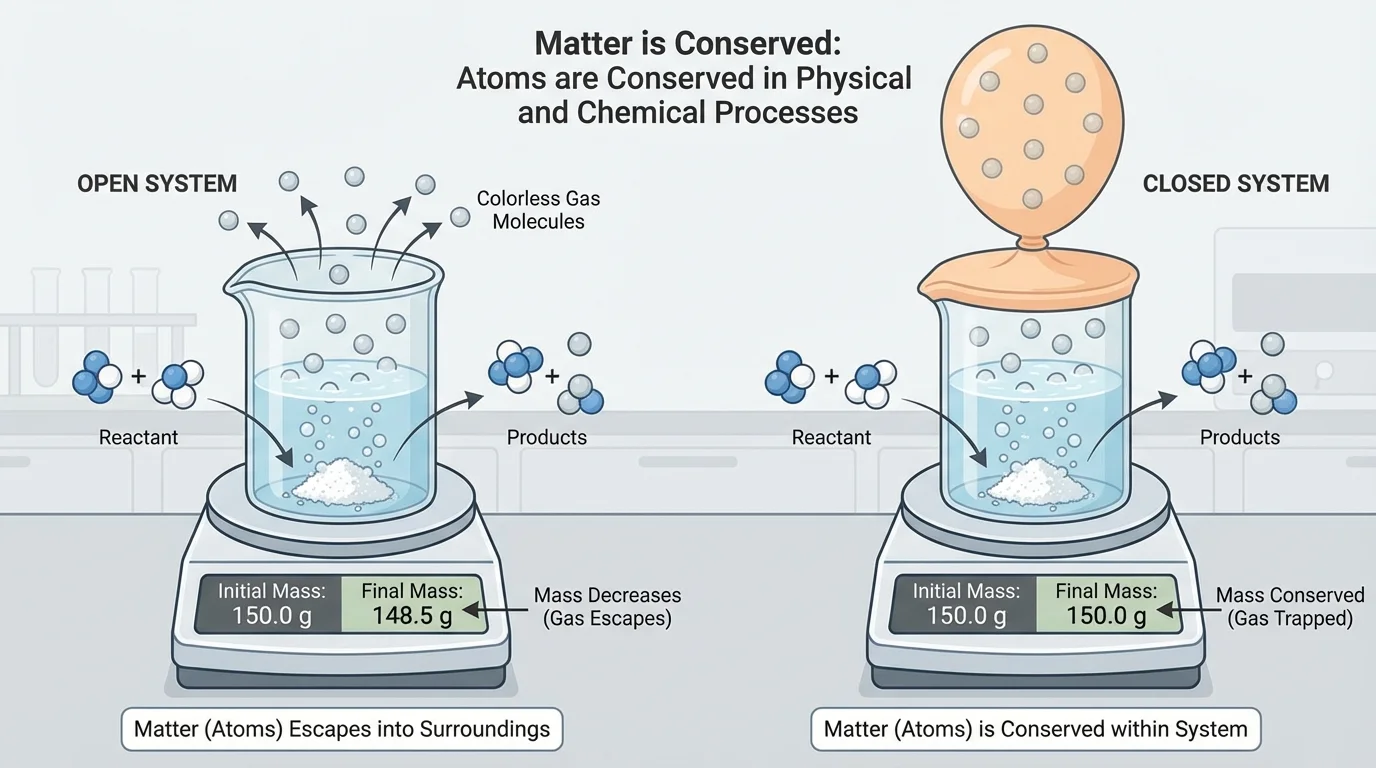
Rusting is another example. Iron combines with oxygen from the air to form rust. The rusted object may even gain mass because oxygen atoms have been added from the air. Again, matter is conserved.
Fizzing tablets or vinegar reacting with baking soda also seem to lose matter if the gas escapes. In a sealed container, the total mass stays constant because the gas remains part of the system.
Mass in an open and closed system
A reaction starts with \(10\) grams of one substance and \(15\) grams of another. The total starting mass is \(25\) grams.
Step 1: Add the reactant masses.
\(10 + 15 = 25\), so the reactants have a total mass of \(25\) grams.
Step 2: Think about conservation of matter.
If no matter enters or leaves, the products must also have a total mass of \(25\) grams.
Step 3: Explain an apparent change.
If the measured mass after the reaction is less in an open container, some product probably escaped as a gas. The matter was not destroyed.
Conservation of matter is easiest to observe when the system is closed.
That is why scientists design experiments carefully. They know that missing matter is often just matter that moved somewhere else.
Conservation of matter matters in medicine, sports, farming, and environmental science. A doctor thinking about nutrition knows that body growth comes from matter entering the body. An athlete who breathes hard during exercise is increasing cellular respiration, turning more glucose and oxygen into carbon dioxide and water.
Farmers and gardeners use this idea too. Compost does not simply vanish as it decomposes. Microorganisms break down the material, and atoms move into the soil, into living cells, and into gases released to the air. The matter changes location and form.
Recycling and pollution also involve conservation of matter. Plastic does not disappear when thrown away. Smoke from engines and factories does not vanish either. The atoms remain in different substances and places, which is why pollution can spread through air, water, soil, and living things.
As we saw in [Figure 4], plants take carbon dioxide from the air and make sugars. This means plants play a major role in the movement of carbon through ecosystems. Understanding this helps scientists study climate, food webs, and the cycling of matter on Earth.
"Atoms are not lost in change; they are rearranged."
Once you understand that idea, many parts of science connect. Food becomes body matter. Breath carries atoms out. Plants pull atoms from air and water. Chemical equations balance because nature keeps track of every atom.