A log burns in a fireplace, and after a while only a small pile of ash remains. At first glance, it looks as if most of the matter simply vanished. But it did not. Most of the atoms from the wood combined with oxygen from the air and became invisible gases such as \(\textrm{CO}_2\) and \(\textrm{H}_2\textrm{O}\). Science often begins by correcting what our eyes suggest, and one of the most powerful corrections is this: in a properly defined system, matter and energy are not lost. They are tracked, transformed, and transferred.
This idea is one of the great organizing principles of chemistry and physics. In chemistry, it explains why a balanced equation must have the same number of each kind of atom on both sides. In physics, it explains why energy changes form rather than disappearing. Together, these ideas let scientists predict what happens in reactions, machines, ecosystems, and even stars.
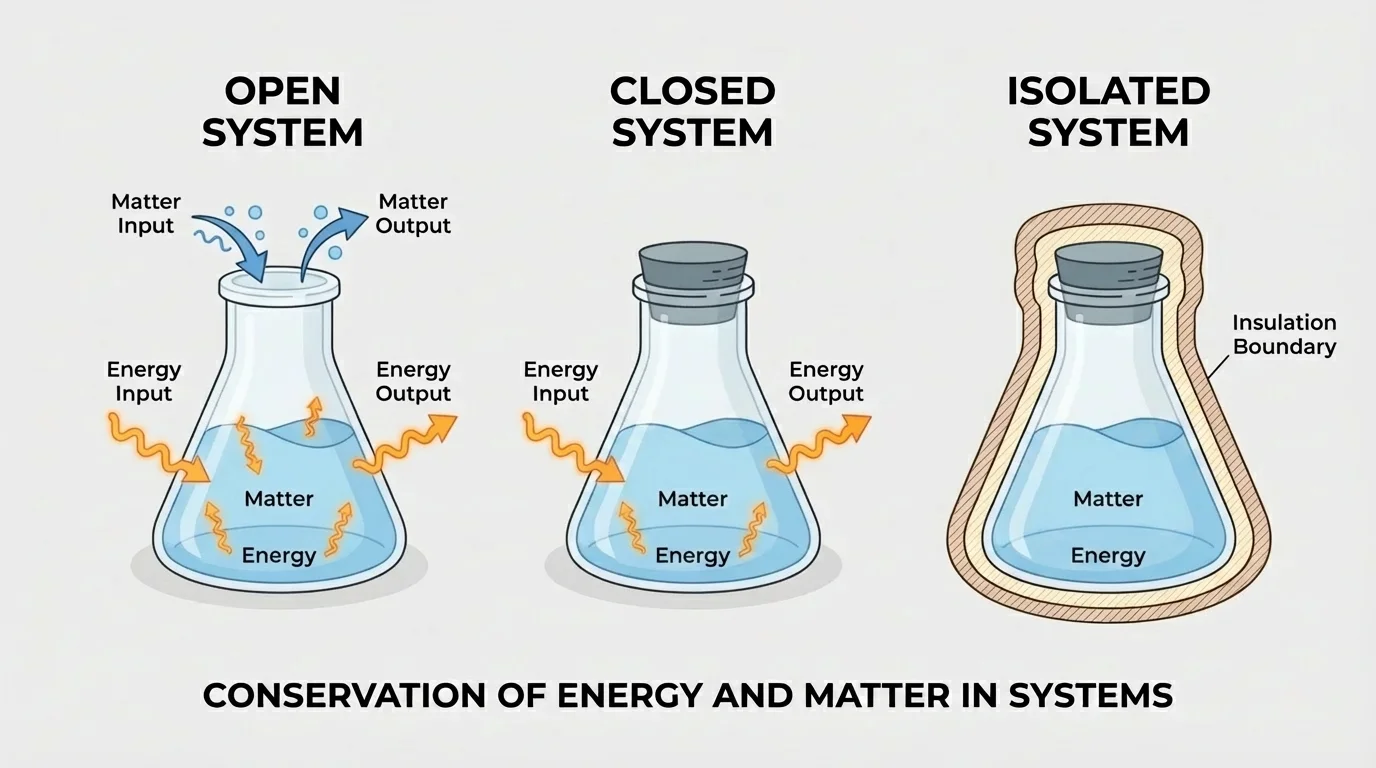
Conservation means that a quantity stays constant overall, even if it changes form or location. A closed system is a system that can exchange energy with its surroundings but not matter. An open system can exchange both matter and energy, while an isolated system exchanges neither.
To decide whether something is conserved, you must first choose the system you are studying and define its boundary clearly. If your boundary is drawn too narrowly, matter or energy may cross it and make conservation seem to fail. When the boundary is chosen correctly, the accounting works.
[Figure 1] Conservation laws are like accounting rules for nature. In financial accounting, money does not disappear without explanation; it is spent, transferred, or stored somewhere else. In science, atoms do not vanish during chemical reactions, and energy does not vanish when motion slows down. Instead, atoms are rearranged and energy is transformed, often into thermal energy that is harder to notice.
This matters because chemistry is fundamentally about rearranging atoms. When methane burns, for example, the atoms in \(\textrm{CH}_4\) and \(\textrm{O}_2\) are reorganized into \(\textrm{CO}_2\) and \(\textrm{H}_2\textrm{O}\). The total count of carbon, hydrogen, and oxygen atoms stays the same. If the equation is not balanced, it does not correctly represent reality.
Conservation also helps explain everyday observations. A phone battery "running out" has not destroyed energy. Chemical potential energy has been transformed into electrical energy, light, sound, and thermal energy. A cooling cup of coffee has not lost energy into nothing; energy has been transferred to the surrounding air, table, and cup.
Whether mass and energy appear to be conserved depends on where you place the boundary for three common kinds of systems. A sealed flask is usually treated as closed because matter stays inside, even though energy can still move in or out as heat. An uncovered beaker is open because gases and liquids may enter or leave.
An isolated system is an ideal model more than a perfect real object. A well-insulated thermos comes close, but in practice a little energy still leaks. Even so, isolated systems are useful for thinking. If neither matter nor energy crosses the boundary, then the total amount inside remains constant in the strongest possible sense.
Suppose you heat water in an open pot. Water vapor escapes, so the mass of the water left in the pot decreases. That does not violate conservation of matter because the pot alone is not a closed system. If you include the steam above the pot and the surrounding air in your system, the matter is still there. The key idea is that conservation depends on tracking all of the matter in the chosen system.
The same is true for energy. A hot metal spoon cools on a table because thermal energy flows into the air and the table. The spoon alone does not conserve its thermal energy, but the larger system consisting of spoon, air, and table does. Nature is consistent; our boundaries determine what we notice.
You already know that matter is made of atoms and that chemical reactions involve breaking old bonds and forming new ones. Keep that idea in mind: the atoms themselves are not created or destroyed in ordinary chemical reactions; only their arrangements change.
[Figure 2] In an ordinary chemical reaction, atoms are rearranged. This is the heart of the law of conservation of mass. The number of atoms of each element before the reaction equals the number after the reaction, as a particle model illustrates. Because atoms are conserved, mass is conserved in a closed system.
Consider the formation of water:
\[2\textrm{H}_2 + \textrm{O}_2 \rightarrow 2\textrm{H}_2\textrm{O}\]
On the left side, there are \(4\) hydrogen atoms and \(2\) oxygen atoms. On the right side, there are also \(4\) hydrogen atoms and \(2\) oxygen atoms. The molecules have changed, but the atom counts match exactly. That is why the equation is balanced.
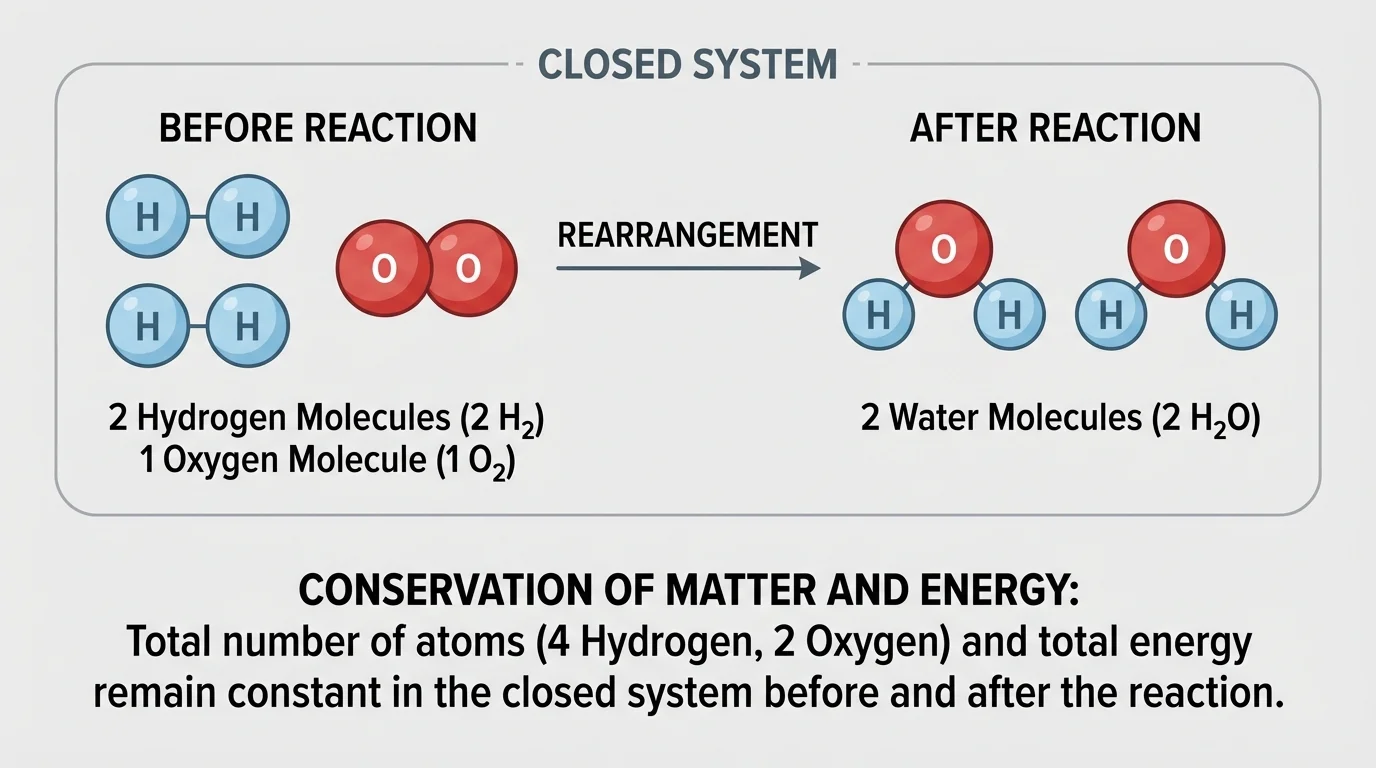
Now consider the burning of methane:
\[\textrm{CH}_4 + 2\textrm{O}_2 \rightarrow \textrm{CO}_2 + 2\textrm{H}_2\textrm{O}\]
Count the atoms. Reactants: carbon \(= 1\), hydrogen \(= 4\), oxygen \(= 4\). Products: carbon \(= 1\), hydrogen \(= 4\), oxygen \(= 4\). Because the atom counts are equal, the equation matches the conservation of matter.
The balanced coefficients are essential. If we wrote \(\textrm{H}_2 + \textrm{O}_2 \rightarrow \textrm{H}_2\textrm{O}\), oxygen would not balance: there would be \(2\) oxygen atoms on the left and only \(1\) on the right. A chemist must adjust coefficients, not subscripts, because changing a subscript changes the identity of the substance.
Atoms are counted, not guessed. A balanced chemical equation is a mathematical representation of conservation. The coefficients tell how many particles or moles of each substance take part. When the equation is correct, each element has the same total number of atoms before and after the reaction.
This particle-level view explains why mass conservation is so reliable in chemistry. Since each atom of carbon, hydrogen, oxygen, sodium, or iron remains present somewhere in the products, the total mass remains constant if no matter escapes. The ash from a fire looks smaller only because much of the product matter has moved into the air.
Chemists do more than count particles in drawings; they also use numbers. The total mass of reactants in a closed system equals the total mass of products. If \(m_{\textrm{reactants}}\) is the mass of all reactants and \(m_{\textrm{products}}\) is the mass of all products, then
\[m_{\textrm{reactants}} = m_{\textrm{products}}\]
This equation is simple, but it becomes powerful when combined with balanced equations and molar masses.
Example 1: Heating iron and sulfur in a sealed container
Iron reacts with sulfur to form iron sulfide:
\[\textrm{Fe} + \textrm{S} \rightarrow \textrm{FeS}\]
Step 1: Record the reactant masses.
Suppose the container holds \(7.0 \textrm{ g}\) of iron and \(4.0 \textrm{ g}\) of sulfur.
Step 2: Add the masses of the reactants.
\(7.0 \textrm{ g} + 4.0 \textrm{ g} = 11.0 \textrm{ g}\)
Step 3: Apply conservation of mass.
If the reaction occurs in a closed container and all reactants form product, the iron sulfide must have mass \(11.0 \textrm{ g}\).
The product mass is \(11.0 \textrm{ g}\).
This seems almost too straightforward, but it captures the main idea: if matter cannot leave, the total mass cannot decrease. If a measurement gives a smaller value, something escaped or the measurement was incomplete.
Example 2: Decomposition of calcium carbonate
In a closed system, calcium carbonate breaks down according to
\[\textrm{CaCO}_3 \rightarrow \textrm{CaO} + \textrm{CO}_2\]
Step 1: Find the total reactant mass.
Suppose \(25.0 \textrm{ g}\) of \(\textrm{CaCO}_3\) decomposes.
Step 2: Use conservation of mass.
The total mass of \(\textrm{CaO}\) and \(\textrm{CO}_2\) together must also be \(25.0 \textrm{ g}\).
Step 3: Solve for an unknown product mass.
If the collected \(\textrm{CO}_2\) has mass \(11.0 \textrm{ g}\), then the \(\textrm{CaO}\) mass is \(25.0 \textrm{ g} - 11.0 \textrm{ g} = 14.0 \textrm{ g}\).
The calcium oxide has mass \(14.0 \textrm{ g}\).
Mole relationships also support conservation. For the reaction \(2\textrm{H}_2 + \textrm{O}_2 \rightarrow 2\textrm{H}_2\textrm{O}\), \(2\) moles of hydrogen molecules react with \(1\) mole of oxygen molecules to produce \(2\) moles of water molecules. The particles are rearranged in a fixed ratio.
Example 3: Mass check with molar masses
Use the reaction
\[2\textrm{H}_2 + \textrm{O}_2 \rightarrow 2\textrm{H}_2\textrm{O}\]
Step 1: Write approximate molar masses.
\(\textrm{H}_2\) is about \(2 \textrm{ g/mol}\), \(\textrm{O}_2\) is about \(32 \textrm{ g/mol}\), and \(\textrm{H}_2\textrm{O}\) is about \(18 \textrm{ g/mol}\).
Step 2: Find the total reactant mass from the coefficients.
\(2\) moles of \(\textrm{H}_2\) have mass \(2 \times 2 = 4 \textrm{ g}\). One mole of \(\textrm{O}_2\) has mass \(32 \textrm{ g}\). Total reactant mass: \(4 \textrm{ g} + 32 \textrm{ g} = 36 \textrm{ g}\).
Step 3: Find the total product mass.
\(2\) moles of \(\textrm{H}_2\textrm{O}\) have mass \(2 \times 18 = 36 \textrm{ g}\).
The masses match: \(36 \textrm{ g} = 36 \textrm{ g}\).
As we saw earlier in [Figure 2], the particle picture and the mass calculation tell the same story from two different scales. One shows atoms; the other shows measurable mass.
Many confusing lab results come from systems that are not actually closed. If a gas escapes, the mass measured on the balance drops. If oxygen from the air enters and reacts with a substance, the mass rises. In both cases, conservation still holds for the larger system.
For example, when magnesium burns in air, it forms magnesium oxide:
\[2\textrm{Mg} + \textrm{O}_2 \rightarrow 2\textrm{MgO}\]
The product can have more mass than the original magnesium strip because oxygen atoms from the air join the solid. Students sometimes think mass was created, but really additional matter entered the system from outside the metal sample.
On the other hand, when baking soda and vinegar react in an open cup, bubbles of \(\textrm{CO}_2\) escape. The cup may seem to lose mass. If the reaction happens in a sealed container, the total mass before and after matches much more closely.
| Situation | What crosses the boundary? | Observed effect | Conservation explanation |
|---|---|---|---|
| Open beaker loses gas | Matter leaves | Measured mass decreases | Total matter is conserved in the larger system |
| Metal burns in air | Matter enters | Measured mass increases | Oxygen becomes part of the product |
| Sealed reaction flask | No matter crosses | Total mass stays constant | Closed-system conservation is directly observed |
Table 1. Common situations in which mass appears to change and the conservation explanation for each case.
Antoine Lavoisier, often called a founder of modern chemistry, used careful mass measurements in closed containers to show that combustion and many other reactions obey conservation of mass. His work helped replace vague ideas about burning with quantitative chemistry.
[Figure 3] Energy conservation is just as important as matter conservation. The first law of thermodynamics states that energy cannot be created or destroyed; it can only be transferred or transformed. A moving object can lose kinetic energy, but that energy becomes some combination of thermal energy, sound, light, or other forms.
Some important forms of energy are kinetic energy, gravitational potential energy, thermal energy, chemical energy, electrical energy, and radiant energy. In a roller coaster, energy shifts from gravitational potential energy at the top to kinetic energy as the car speeds downward. Because friction is present, some energy also becomes thermal energy in the wheels, track, and air.
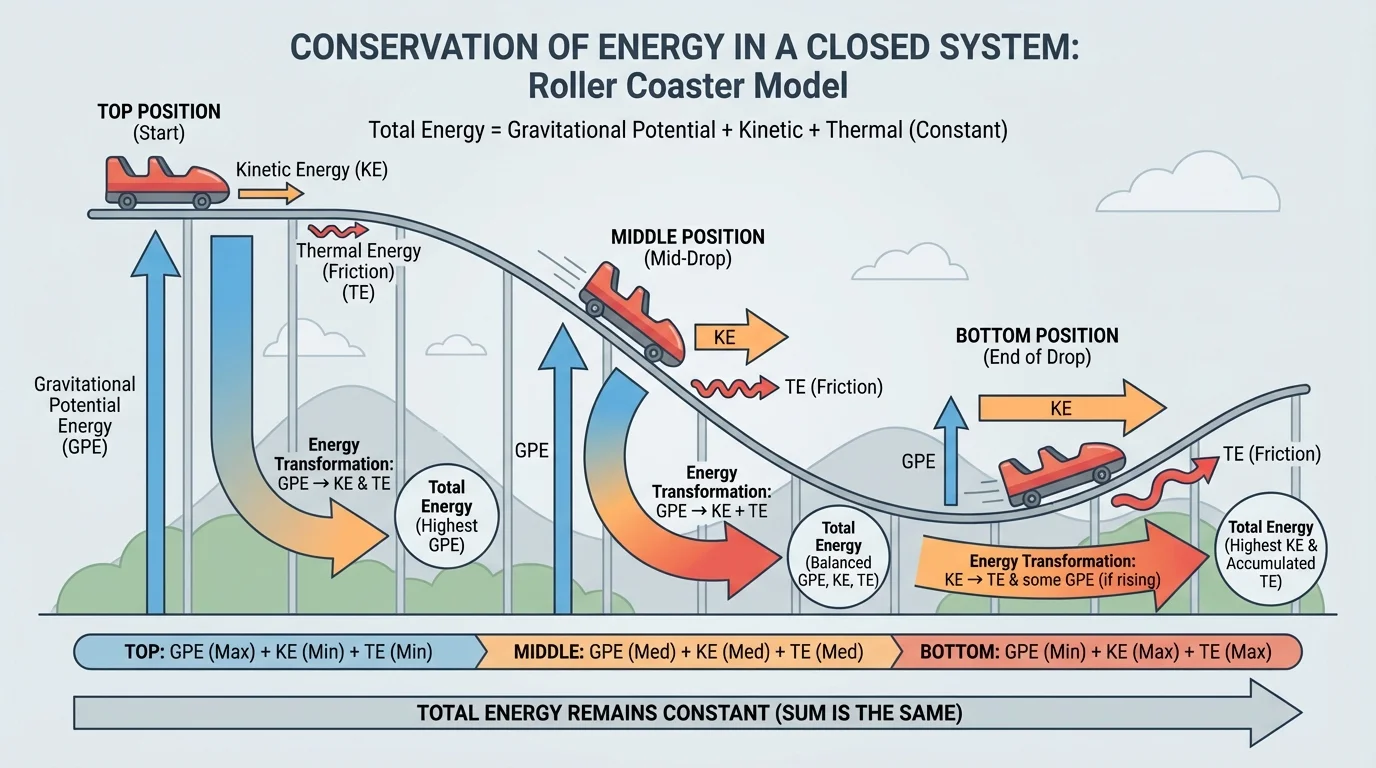
In an ideal frictionless case, the total mechanical energy would remain constant. In real life, mechanical energy decreases, but total energy still stays constant because the "missing" mechanical energy is transformed into thermal energy and sound. This is why brakes get hot and why your hands warm up when you rub them together.
A common mathematical statement of energy conservation in a closed system is
\[E_{\textrm{initial}} = E_{\textrm{final}}\]
For thermal energy transfer, scientists often use
\[q = mc\Delta T\]
where \(q\) is thermal energy transferred, \(m\) is mass, \(c\) is specific heat capacity, and \(\Delta T\) is temperature change.
Suppose \(100 \textrm{ g}\) of water warms by \(5.0^\circ\textrm{C}\). Using \(c = 4.18 \textrm{ J/(g}\cdot\textrm{ ^\circ C)}\), the thermal energy gained is \(q = 100 \times 4.18 \times 5.0 = 2{,}090 \textrm{ J}\). That energy came from somewhere else in the system, such as a hot metal object or a heater.
"Energy cannot be created or destroyed, only transformed from one form to another."
— First Law of Thermodynamics
In most high school chemistry, matter conservation means atoms are conserved during chemical reactions. That is an excellent approximation and the correct model for balancing equations and solving reaction-mass problems. Chemical reactions rearrange electrons and bonds, but they do not turn significant amounts of matter into energy.
At a deeper level, modern physics connects mass and energy through \(E = mc^2\). This relation becomes especially important in nuclear reactions, where tiny changes in mass correspond to very large energy changes. In ordinary chemical reactions, however, the mass change is so small that it is usually far below what classroom measurements can detect.
This distinction matters. For chemistry problems involving equations such as \(\textrm{NaOH} + \textrm{HCl} \rightarrow \textrm{NaCl} + \textrm{H}_2\textrm{O}\), you should conserve atoms and mass directly. For nuclear processes such as fusion in stars, the mass-energy relationship becomes central.
Chemical change versus nuclear change. In chemical reactions, atoms keep their identities and are rearranged into new substances. In nuclear reactions, the nuclei themselves change, so matter and energy must be considered together more explicitly.
Conservation laws are not just abstract ideas; they are used constantly in engineering, environmental science, and medicine. In car engines, fuel and oxygen react to form exhaust gases, and the chemical energy of the fuel is transformed into motion and thermal energy. Engineers track both mass flow and energy efficiency to design better engines.
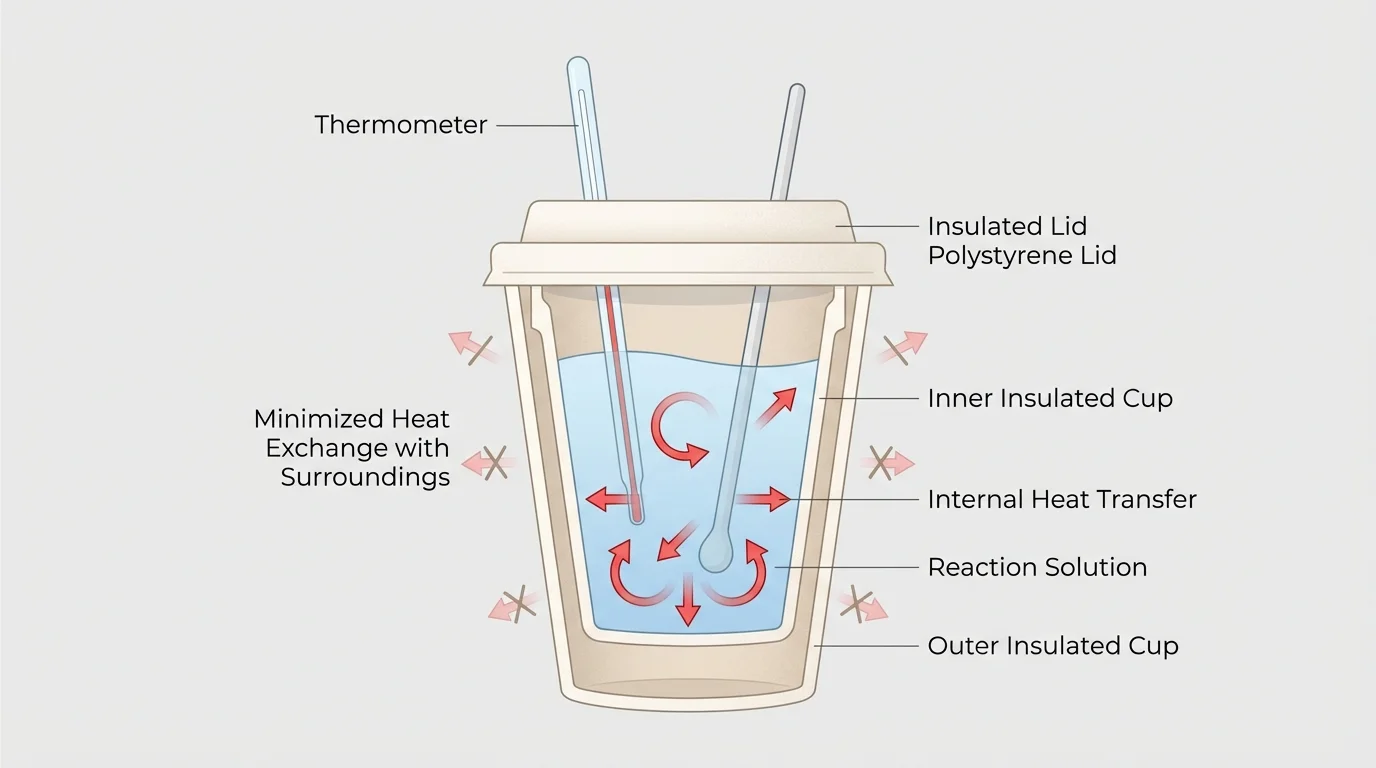
[Figure 4] In chemical manufacturing, every atom has financial value. If a plant starts with a certain mass of raw materials, managers want to know exactly how much desired product, waste, and emissions result. A calorimeter is a device that helps measure energy changes by tracking temperature changes in a controlled setup. This allows chemists to connect observable temperature change to energy transfer.
In a simple coffee-cup calorimeter, if one substance warms while another cools, the energy gained by one part is approximately equal to the energy lost by another, provided the setup is well insulated. The calculation often uses \(q = mc\Delta T\), and the signs of the energy values indicate gain or loss.
Environmental science also depends on conservation. Carbon atoms from gasoline do not disappear after combustion; they enter the atmosphere mainly as \(\textrm{CO}_2\). This is one reason carbon accounting matters in climate science. The same conservation principle used to balance a classroom equation also helps scientists track greenhouse gases globally.
In biology and medicine, metabolism follows the same rules. Food contains chemical energy. Your body transforms that energy into motion, thermal energy, electrical signaling in nerves, and stored molecules. The atoms from nutrients become part of body tissues, exhaled \(\textrm{CO}_2\), water, and waste products. Conservation provides the bookkeeping.
Later, when studying reaction energetics, the calorimetry setup in [Figure 4] remains useful because it makes invisible energy transfer measurable through temperature. In the same way, the roller coaster picture from [Figure 3] reminds us that energy often changes form rather than disappearing from the system.
A sealed plastic bag containing vinegar and baking soda can demonstrate conservation of mass clearly. Before mixing, the bag and contents have a certain mass. After the reaction produces \(\textrm{CO}_2\), the bag inflates, but the total mass of the sealed bag stays essentially the same because no matter leaves.
Another demonstration uses steel wool. If steel wool is weighed before and after reacting with oxygen in air, its mass increases. The increase makes sense only when oxygen crossing the boundary is included in the system description. This is a powerful reminder that boundaries matter as much as equations.
A thermal demonstration can use warm and cool water in an insulated container. The warm water loses energy, the cool water gains energy, and the final temperature reflects energy redistribution within the system. No energy is created; it is transferred until the system moves toward thermal equilibrium.