A cup of hot chocolate and a swimming pool can both be at the same temperature, but no one would say they contain the same amount of thermal energy. That surprising fact shows why science often needs more than one measurement at a time. To understand what is really happening, scientists compare quantities using ratios and proportional relationships. Just as speed compares distance and time, heating compares energy transferred, mass, and temperature change.
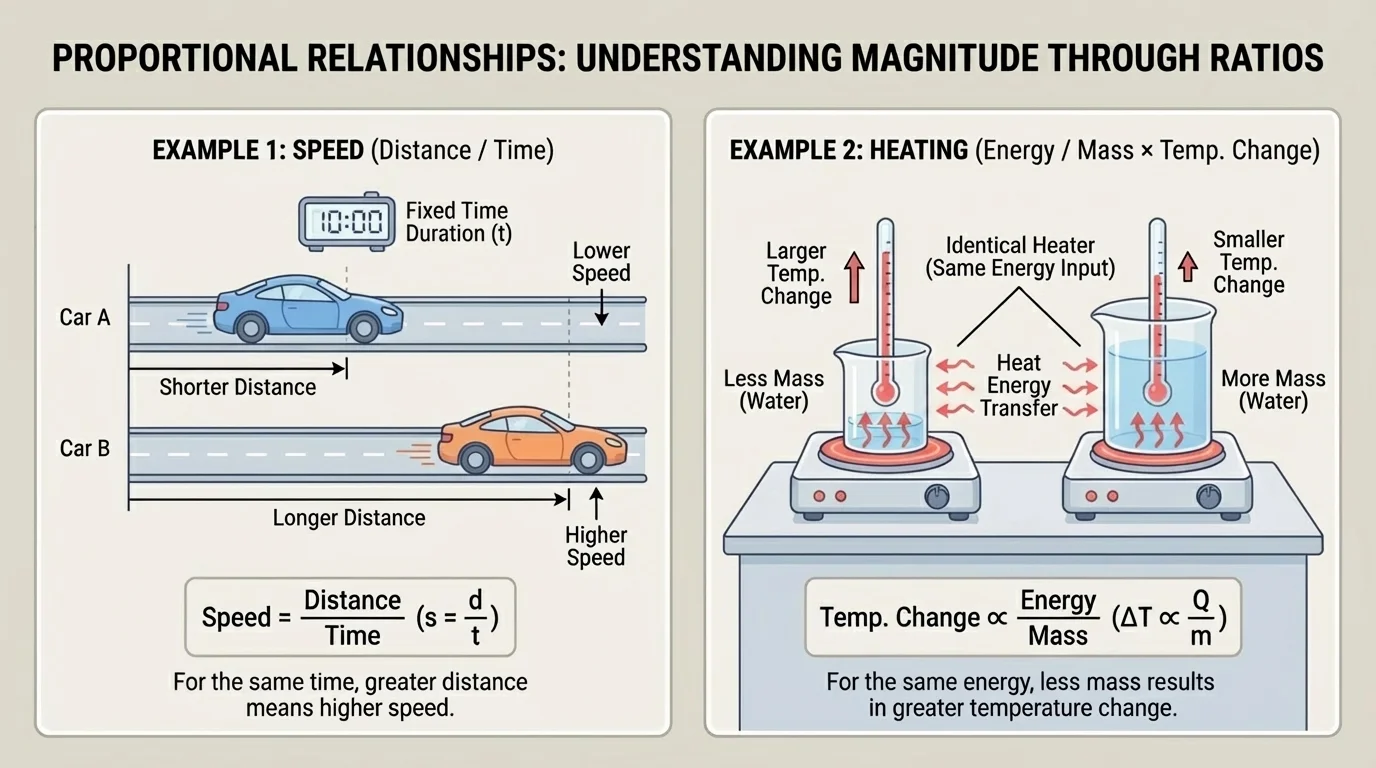
A proportional relationship happens when two quantities change together in a predictable way. [Figure 1] shows how scientists use these relationships to understand both motion and heating. For example, speed tells how much distance is traveled in a certain amount of time. If a skateboarder travels farther in the same amount of time, the speed is greater.
Speed can be written as a ratio:
\[\textrm{speed} = \frac{\textrm{distance}}{\textrm{time}}\]
If a cyclist travels \(24 \textrm{ km}\) in \(2 \textrm{ h}\), then the speed is \(\dfrac{24}{2} = 12 \textrm{ km/h}\). This ratio gives information about the magnitude of motion. In the same way, scientists use ratios and relationships to understand the magnitude of heating, cooling, and energy transfer.
When energy is transferred to matter, the particles in that matter usually move faster on average. Faster particle motion means greater average kinetic energy, and that is measured by temperature. But the size of the temperature change depends on more than just the energy added. It also depends on how much matter there is and what kind of matter it is.
You already know that matter is made of tiny particles and that temperature is related to how fast those particles move on average. You also know that energy can be transferred from one place to another, such as from a stove to a pan or from sunlight to sand.
Proportional thinking helps connect those ideas. Instead of just saying that one sample got hotter, we can ask how much hotter, how much energy was transferred, and how much material was heated. Those comparisons make science more precise.
In a heating investigation, several important quantities are measured. The first is energy transfer, which means energy moving from one object or system to another. If a hot plate warms water, energy moves from the hot plate into the water.
The second is mass, which is the amount of matter in a sample. A larger mass has more particles, so the same amount of added energy is shared among more particles.
The third is temperature, which measures the average kinetic energy of the particles in a sample. If the particles move faster on average, the temperature rises.
The fourth quantity is the type of matter. Water, sand, cooking oil, and aluminum do not all respond to heating in the same way. Some substances need more energy than others to raise their temperature by the same amount.
Average kinetic energy is the average energy of motion of the particles in a substance. Temperature is a measure of that average kinetic energy. When temperature increases, the particles are moving faster on average.
These quantities are connected. If you keep the material the same and heat a small sample and a large sample with equal energy, the smaller sample usually has the bigger temperature increase. If you keep the mass the same but change the material, the temperature increase may still be different.
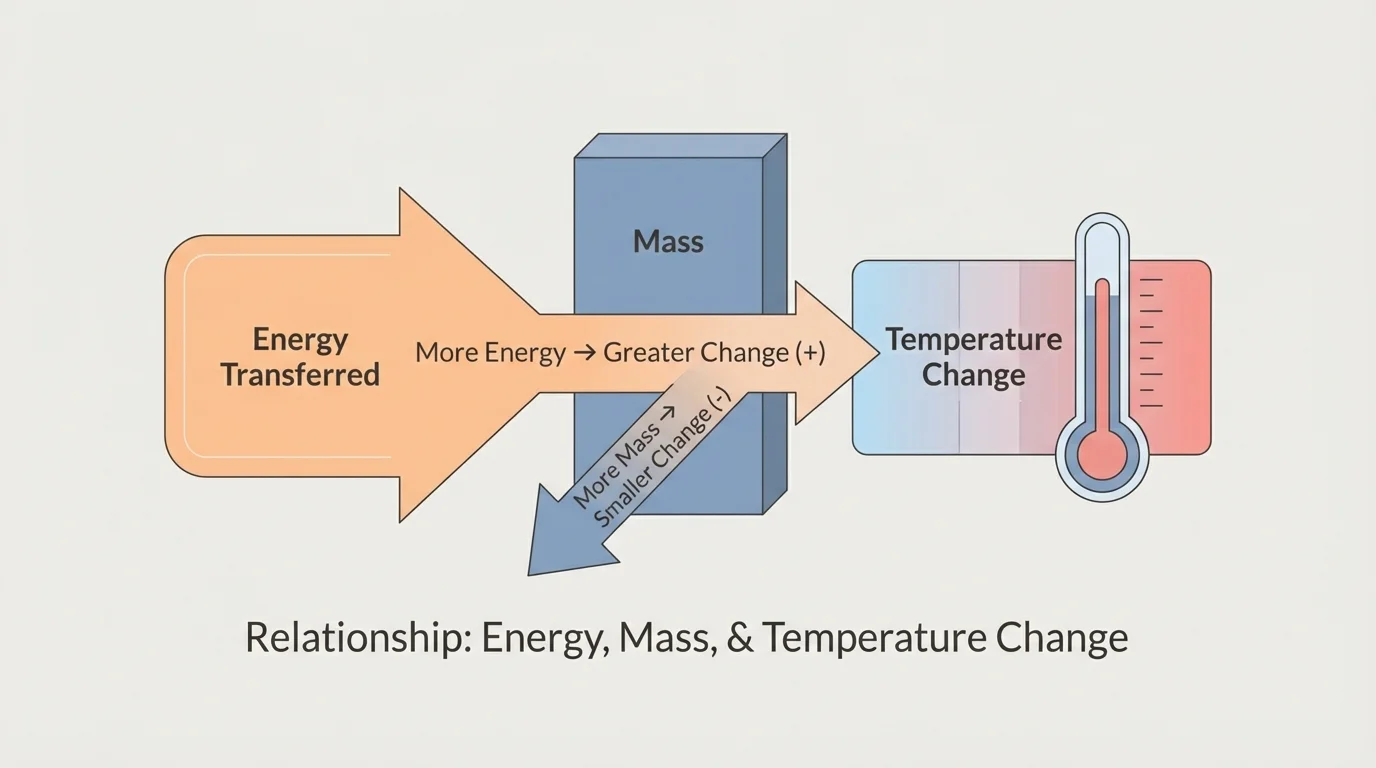
[Figure 2] helps show that in science, some quantities are directly related, and some are inversely related. A direct relationship means that as one quantity increases, the other also increases in a consistent way. An inverse relationship means that as one quantity increases, the other decreases in a consistent way.
Suppose the type of matter and the mass stay the same. If you transfer more energy to the sample, the temperature change becomes larger. For that situation, energy transferred and temperature change have a direct relationship.
Now suppose the type of matter and the energy transferred stay the same. If the mass becomes larger, the temperature change becomes smaller. For that situation, mass and temperature change have an inverse relationship.
This makes sense if you think about particles. Adding energy to a small sample means fewer particles share that energy. Adding the same energy to a larger sample means more particles share it, so the average increase in particle motion is smaller.
For example, if a heater transfers the same amount of energy to \(100 \textrm{ g}\) of water and to \(200 \textrm{ g}\) of water, the \(100 \textrm{ g}\) sample will have the larger temperature increase. If the larger mass is about twice as much, the temperature change is often about half as much, as long as the material stays the same and energy losses are small.
On a sunny day, dry sand at the beach can feel much hotter than ocean water nearby even though both receive energy from the Sun. The difference happens because the two materials respond differently to the same energy input.
The same idea appears in other proportional situations. If a car keeps a steady speed, doubling the travel time doubles the distance. In heating, if the material and mass stay fixed, doubling the energy transferred approximately doubles the temperature change.
Scientists often summarize the relationship among these quantities with an equation:
\[Q = mc\Delta T\]
In this relationship, \(Q\) is the energy transferred, \(m\) is the mass, \(c\) is a number that depends on the material, and \(\Delta T\) means the change in temperature. The symbol \(c\) is called specific heat capacity, which tells how much energy is needed to raise the temperature of a certain mass of a substance by a certain amount.
You do not need to memorize difficult units to understand the idea. What matters most is the pattern. If \(m\) and \(c\) stay the same, then larger \(Q\) gives larger \(\Delta T\). If \(Q\) and \(c\) stay the same, then larger \(m\) gives smaller \(\Delta T\).
Worked example: same material, different energy
A metal sample has the same mass each time it is heated. In Trial A, it receives \(100\) units of energy and its temperature rises by \(5^\circ \textrm{C}\). In Trial B, it receives \(200\) units of energy.
Step 1: Identify the relationship.
Because the mass and type of matter stay the same, energy transferred and temperature change are directly related.
Step 2: Compare the energy amounts.
Trial B has \(\dfrac{200}{100} = 2\) times as much energy as Trial A.
Step 3: Scale the temperature change.
The temperature change is also multiplied by \(2\): \(5 \times 2 = 10\).
The temperature rise in Trial B is \(10^\circ \textrm{C}\).
That example shows a proportional relationship. The ratio \(\dfrac{Q}{\Delta T}\) stays constant when the mass and type of matter stay constant.
Here is another way to think about it. If \(Q = mc\Delta T\), then for one material and one mass, \(mc\) is constant. That means \(Q\) and \(\Delta T\) rise together in a predictable way.
Worked example: same energy, different mass
A heater transfers the same energy to two water samples. Sample A has mass \(100 \textrm{ g}\) and warms by \(8^\circ \textrm{C}\). Sample B has mass \(200 \textrm{ g}\).
Step 1: Identify what stays the same.
The energy transferred and the material are the same.
Step 2: Compare the masses.
Sample B has \(\dfrac{200}{100} = 2\) times the mass of Sample A.
Step 3: Apply the inverse relationship.
If mass doubles, temperature change is cut in half: \(\dfrac{8}{2} = 4\).
Sample B warms by \(4^\circ \textrm{C}\).
Real investigations are not always perfectly proportional because some energy can escape into the air or container. Even so, the equation helps explain the main trend.
[Figure 3] shows that the type of matter has a huge effect on temperature change. Water usually needs more energy than many other common substances to increase its temperature by the same amount. Metals often warm up quickly. Sand can also heat faster than water under the same sunlight.
If equal masses of water, sand, and aluminum each receive the same energy, they usually end with different temperature increases. That does not mean energy disappeared or appeared from nowhere. It means each material stores transferred energy differently at the particle level.
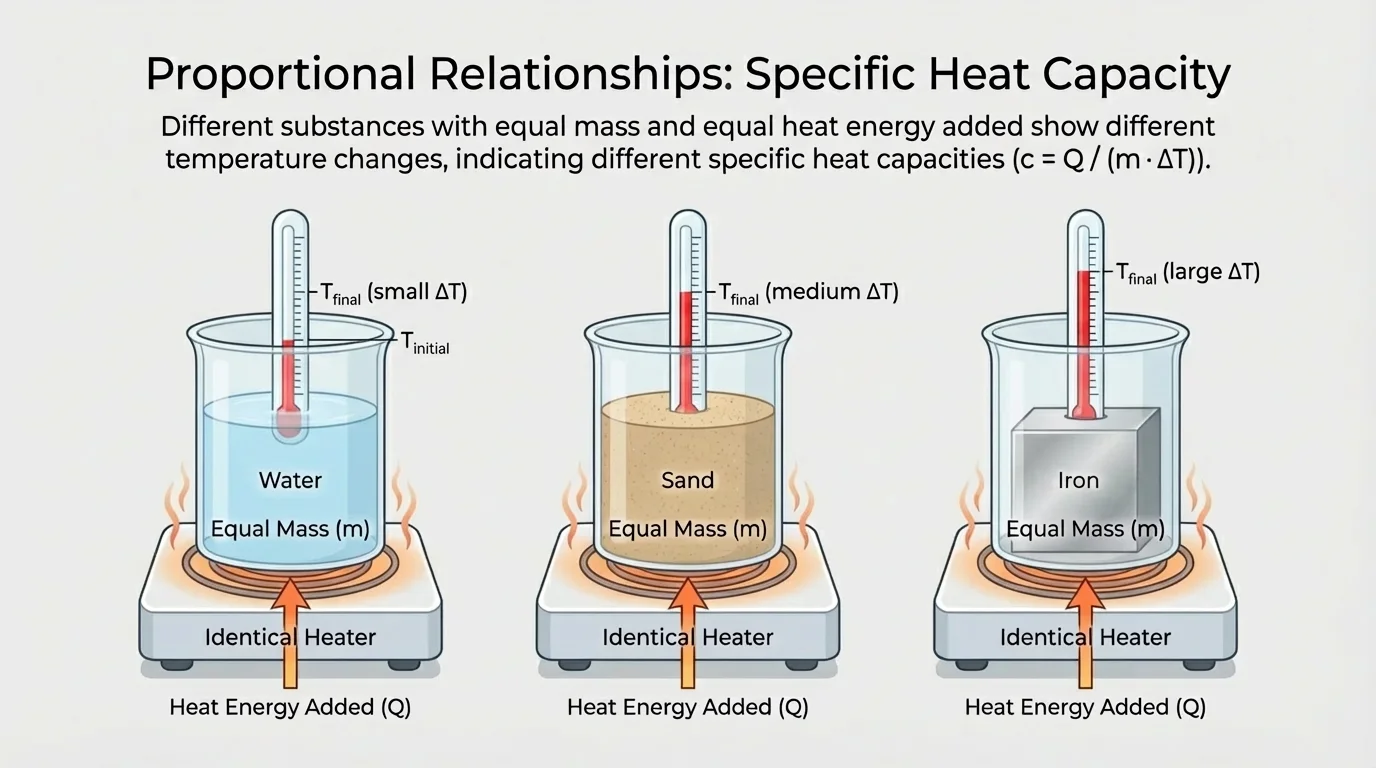
This is why beaches can have hot sand and cooler water during the day. The Sun transfers energy to both, but the water's temperature changes less for the same energy because water has a higher specific heat.
It also helps explain cookware. A metal pan can heat quickly on a stove because the material responds strongly to energy transfer. A pot of water on that stove often takes longer to rise in temperature because the water needs more energy for each degree of warming.
Worked example: comparing materials
Two samples each have mass \(100 \textrm{ g}\). One is water and one is sand. The same energy is transferred to both. The sand warms by \(12^\circ \textrm{C}\), while the water warms by \(5^\circ \textrm{C}\).
Step 1: Identify what is the same.
The energy transferred and the mass are the same.
Step 2: Identify what is different.
The type of matter is different.
Step 3: Interpret the result.
Because the water's temperature changes less, water requires more energy per degree of warming than sand does.
The material itself affects how much the temperature changes.
Looking back at [Figure 1], this is similar to how two trips can have different speeds even if the time is the same. The ratio tells important information about the process.
[Figure 4] shows one way to investigate heating scientifically. You need a fair test, which changes one factor at a time while keeping the others controlled. In a heating experiment, you might measure mass with a balance, time with a stopwatch, and temperature with a thermometer or digital sensor.
If you want to test how mass affects temperature change, you should keep the type of matter the same, use the same heating device, and transfer energy for the same amount of time. If you want to test how the type of matter affects temperature change, you should keep the mass the same and use equal energy transfer for each sample.
Scientists call the factor being changed the variable. Other conditions that are kept the same are called controlled variables. Careful control is important because if several things change at once, it becomes hard to know which one caused the result.
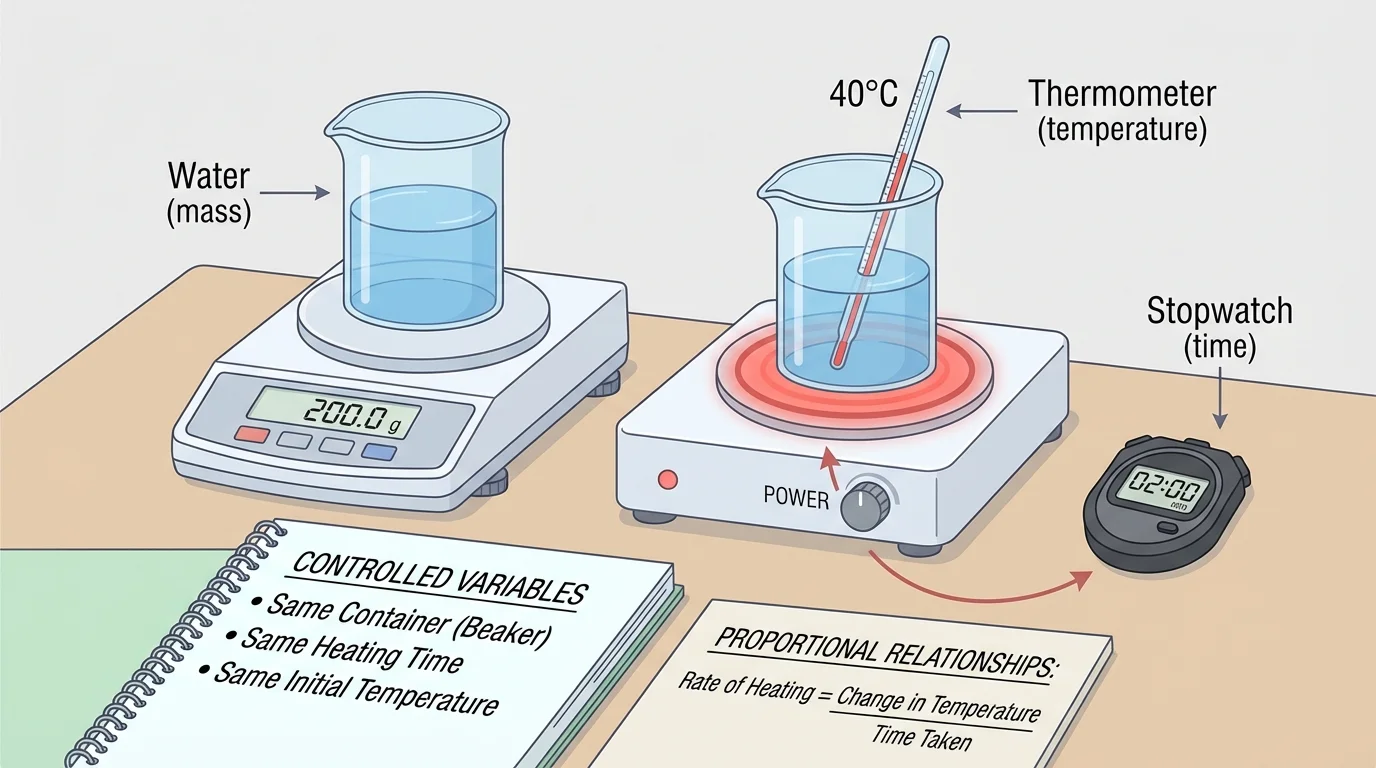
A simple data table can help organize results.
| Trial | Material | Mass | Heating time | Initial temperature | Final temperature | Temperature change |
|---|---|---|---|---|---|---|
| \(1\) | Water | \(100 \textrm{ g}\) | \(2 \textrm{ min}\) | \(20^\circ \textrm{C}\) | \(26^\circ \textrm{C}\) | \(6^\circ \textrm{C}\) |
| \(2\) | Water | \(200 \textrm{ g}\) | \(2 \textrm{ min}\) | \(20^\circ \textrm{C}\) | \(23^\circ \textrm{C}\) | \(3^\circ \textrm{C}\) |
| \(3\) | Sand | \(100 \textrm{ g}\) | \(2 \textrm{ min}\) | \(20^\circ \textrm{C}\) | \(32^\circ \textrm{C}\) | \(12^\circ \textrm{C}\) |
Table 1. Example data from a heating investigation comparing mass and type of matter.
From this table, you can compare ratios and patterns. Trial \(2\) has twice the mass of Trial \(1\) and about half the temperature change. Trial \(3\) has the same mass as Trial \(1\) but a larger temperature change because the material is different.
What makes an investigation reliable? Reliable investigations use careful measurements, repeated trials, and controlled conditions. If you repeat the same test several times and get similar results, you can trust the pattern more strongly.
Investigators also need to think about safety. Hot plates, warm liquids, and glass containers must be handled carefully. Accuracy matters too: reading thermometers at eye level and measuring mass correctly improve the quality of the data.
[Figure 5] shows one common pattern that helps scientists see proportional relationships. If the same material and mass are used each time, a graph of energy transferred versus temperature change often forms a straight-line trend through the origin or very near it.
That straight-line pattern means the quantities are proportional. For example, if \(50\) units of energy cause a \(2^\circ \textrm{C}\) rise, then \(100\) units may cause about a \(4^\circ \textrm{C}\) rise, and \(150\) units may cause about a \(6^\circ \textrm{C}\) rise.
The ratio stays nearly constant:
\[\frac{50}{2} = 25, \quad \frac{100}{4} = 25, \quad \frac{150}{6} = 25\]
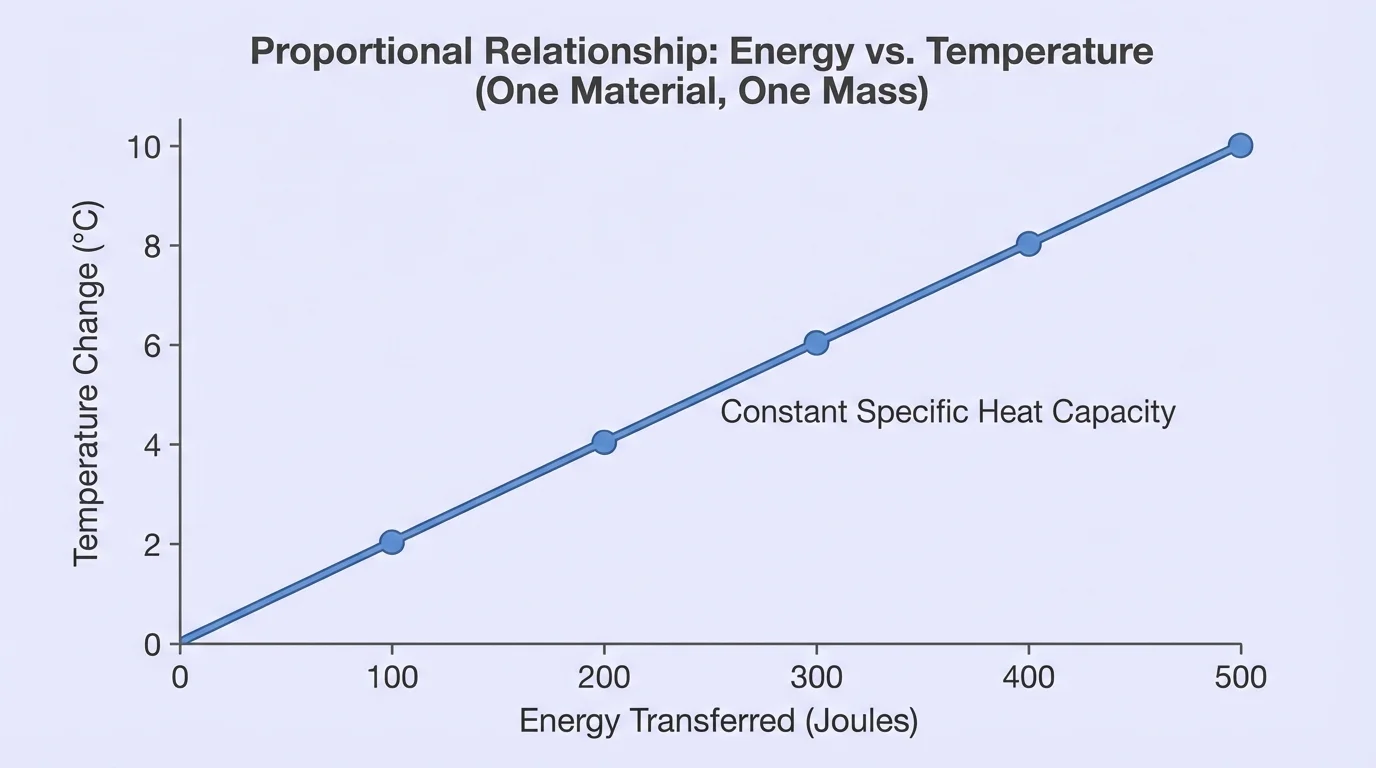
But not every graph in heating is proportional. If the material changes from trial to trial, or if heat escapes a lot, the graph may not be a straight line. Scientists must decide whether the pattern is truly proportional or only approximately so.
A graph can also help compare materials. If one material's line is steeper than another's on a graph of temperature change versus energy, that material's temperature changes more for each unit of transferred energy. Connecting this to [Figure 3], steeper change often belongs to materials that heat up more quickly.
These relationships are not just for lab class. They explain many things students notice every day. Cooking is one example. Heating soup in a small bowl is faster than heating the same soup in a giant pot because the smaller mass usually needs less energy to change temperature by the same amount.
Weather and climate also connect to this idea. Large bodies of water warm and cool more slowly than land. Because water changes temperature more slowly, coastal areas often have milder temperatures than inland areas.
Engineers use these relationships when choosing materials for buildings, machines, and electronics. A device that gets hot quickly may need materials that can withstand or dissipate heat safely. Athletes also experience this in hot weather because water in the body helps reduce sudden temperature changes.
"The right measurement can turn a surprising event into an understandable pattern."
Medicine uses the same thinking. A warm compress transfers energy to body tissue, but the temperature change depends on the tissue, the mass of the area being warmed, and how long the energy is transferred. Scientists and doctors need these relationships to be predictable and safe.
One common mistake is thinking that higher temperature always means more total energy. That is not true. A bathtub of warm water can contain more total thermal energy than a cup of very hot tea because the bathtub has much more mass.
Another mistake is confusing temperature change with energy transferred. If two samples change by different amounts, the cause might be a different mass or a different material, not just a different amount of energy. Looking again at [Figure 2], the relationship depends on which quantities are held constant.
It is also important to remember that proportional relationships work best under controlled conditions. If water starts boiling or a material melts, the relationship may change because the transferred energy is being used in new ways. Science works by noticing where a pattern holds and where it stops holding.
When you study proportional relationships in energy, you are learning a powerful scientific habit: compare quantities carefully, keep track of what stays the same, and use data to explain what happens. That is how scientists turn observations into evidence.