Every second, Earth is flooded with sunlight, yet a beam of light cannot directly power your muscles, build a tree trunk, or become the paper in a notebook. For solar energy to become useful to living systems, it must be captured and converted into a stored form. The main way this happens on Earth is through photosynthesis, a complex chemical process that turns radiant energy from the Sun into chemical energy stored in matter.
This idea is one of the most important connections between matter and energy in science. Plants, algae, and certain bacteria do not just "use" sunlight. They transform it. They take relatively low-energy raw materials from the environment, especially the gas \(\textrm{CO}_2\) and water \(\textrm{H}_2\textrm{O}\), and build energy-rich organic molecules such as glucose, \(\textrm{C}_6\textrm{H}_{12}\textrm{O}_6\). That stored chemical energy later moves through ecosystems, fuels metabolism, and even, over geologic time, contributes to fossil fuels.
Sunlight is a form of radiant energy, but most organisms cannot store light directly. Energy must be converted into a form that can be held in chemical bonds and used later. Think of sunlight like income paid in a currency that cells cannot spend directly. Photosynthesis acts like the exchange system that converts that energy into a biological "savings account."
When you eat fruit, grains, or meat, you are ultimately using energy that was first captured by photosynthetic organisms. Even when a wolf eats a deer, the energy in the wolf's body originally came from plants that captured sunlight. This is why photosynthesis sits near the base of almost every food web on Earth.
Energy can change form but is not created from nothing. In photosynthesis, radiant energy from sunlight is transformed into chemical energy stored in bonds within organic molecules.
That transformation matters because chemical energy can be stored, moved, and released later when cells need it. A leaf exposed to sunlight can build sugars during the day, and those sugars can later be used for growth, repair, transport, and reproduction.
Autotrophs are organisms that can make their own organic molecules from inorganic substances. Most familiar autotrophs are plants, but algae and some bacteria also carry out photosynthesis.
Photosynthesis is the process by which certain organisms use light energy to convert carbon dioxide and water into sugars and other organic molecules, releasing oxygen as a byproduct in many forms of the process.
Chemical energy is energy stored in the arrangement of atoms and bonds within molecules.
Radiant energy is energy carried by electromagnetic waves, including visible light from the Sun.
The overall simplified equation for photosynthesis is:
\[6\textrm{CO}_2 + 6\textrm{H}_2\textrm{O} + \textrm{light energy} \rightarrow \textrm{C}_6\textrm{H}_{12}\textrm{O}_6 + 6\textrm{O}_2\]
This equation is useful, but it hides a lot of complexity. Photosynthesis is not a single reaction. It is a network of many linked reactions, enzymes, membranes, pigments, and electron transfers. Still, the equation captures the big picture: carbon dioxide and water are rearranged using light energy to produce sugar and oxygen.
Notice that the oxygen gas released to the atmosphere comes from water molecules that are split during the process, not directly from carbon dioxide. That detail was a major scientific discovery because it revealed how deeply matter is reorganized during photosynthesis.
In plants, photosynthesis depends on specialized structures inside leaves and cells, as [Figure 1] shows. Leaves are shaped to capture light efficiently, while tiny pores called stomata allow gases such as carbon dioxide and oxygen to move in and out.
Inside many leaf cells are organelles called chloroplasts. These are the main sites of photosynthesis in plants and algae. Within chloroplasts are stacks of membrane sacs called thylakoids, surrounded by a fluid region called the stroma.
The green pigment chlorophyll is embedded in the thylakoid membranes. Chlorophyll absorbs some wavelengths of visible light especially well, particularly red and blue light, while reflecting more green light, which is why most plant leaves look green.
Structure matters because energy capture is not random. The arrangement of pigments and membranes helps organisms absorb light, move electrons, and build the molecules needed for the next stage of the process. Later, when considering why some conditions speed up photosynthesis and others slow it down, it helps to remember the structural organization shown earlier in [Figure 1].
Photosynthesis is usually described in two major stages, as [Figure 2] illustrates: the light-dependent reactions and the Calvin cycle. These stages are linked by both matter and energy.
In the light-dependent reactions, chlorophyll and related pigments absorb photons of light. This excites electrons, raising their energy. Those energized electrons move through an electron transport system in the thylakoid membrane. Their movement helps produce ATP and NADPH, two molecules that temporarily store usable energy for the cell.
Water is also split during this stage. This process provides replacement electrons and hydrogen ions, and it releases oxygen gas. The basic idea can be represented simply as light energy driving the breakdown of \(\textrm{H}_2\textrm{O}\) and the formation of energy-carrying molecules. Oxygen produced here diffuses out of the leaf and eventually enters the atmosphere.
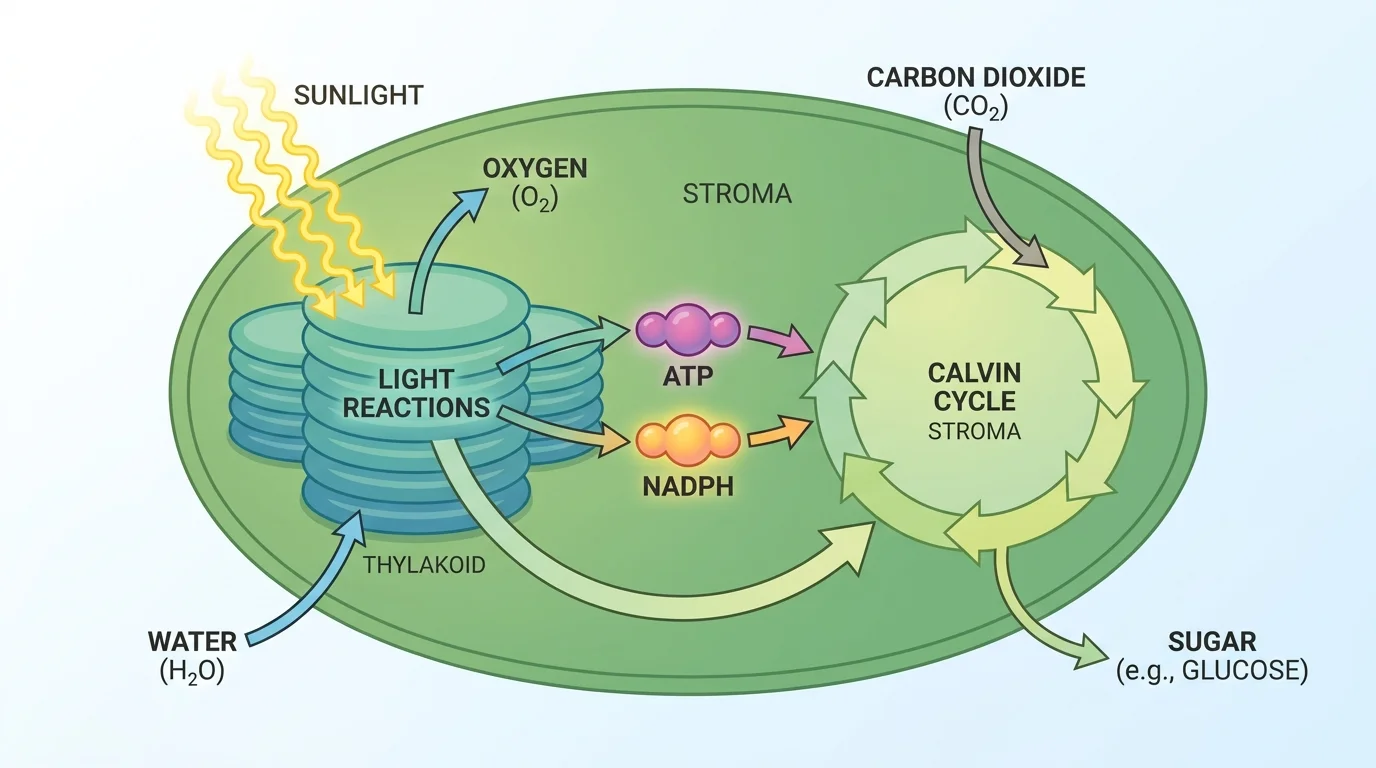
In the Calvin cycle, which takes place in the stroma, carbon dioxide is incorporated into organic molecules. This process is often called carbon fixation because inorganic carbon from \(\textrm{CO}_2\) becomes part of carbon-containing organic compounds. ATP and NADPH from the light-dependent reactions provide the energy and reducing power needed to build sugars.
Why two stages? Light energy is difficult to store directly in a stable form. The first stage captures light and transfers its energy into short-term chemical carriers such as ATP and NADPH. The second stage uses those carriers to assemble energy-rich carbon compounds. This organization makes photosynthesis efficient and controllable.
A simplified way to think about it is this: the first stage captures energy, and the second stage uses that energy to build sugar. Even though students often memorize "plants make glucose," the real achievement is that sunlight energy becomes locked into the bonds of carbon-based molecules.
ATP is not the long-term storage product of photosynthesis. It acts more like a rechargeable battery used immediately inside the chloroplast. Long-term storage is mainly found in sugars and other organic molecules that can be transported or converted into starches, oils, and structural materials.
The most important idea in this topic is not just that plants make organic molecules. It is that matter and energy are both transformed in a coordinated way. The atoms in glucose come mostly from carbon dioxide and water, while the energy stored in glucose comes from sunlight.
When scientists say energy is "stored" in glucose, they mean that the molecule's chemical structure contains bonds that can later be rearranged in reactions such as cellular respiration. During respiration, cells break down glucose and release some of that stored energy in a controlled way.
The key transformation can be described like this: radiant energy \(\rightarrow\) chemical energy. In photosynthesis, this transformation depends on many smaller transfers involving electrons. Electrons absorb energy from light, move through carrier molecules, and help drive the synthesis of larger organic compounds.
Here is a simple numerical way to think about storage. Suppose a plant leaf produces \(10\) grams of glucose over time. That new glucose represents matter added to the plant and chemical energy stored in that matter. The leaf has not created matter or energy from nothing. It has rearranged atoms from \(\textrm{CO}_2\) and \(\textrm{H}_2\textrm{O}\) and transformed incoming light energy into a form that can be stored.
Energy storage example
Biologists often estimate the chemical energy in glucose at about \(2{,}870\ \textrm{kJ}\) per mole when fully oxidized.
Step 1: Identify the amount of glucose
One mole of glucose has a mass of about \(180\ \textrm{g}\). If a plant makes \(18\ \textrm{g}\) of glucose, that is \(0.1\ \textrm{mol}\).
Step 2: Calculate stored chemical energy
\[0.1 \times 2870 = 287 \textrm{ kJ}\]
This means \(18\ \textrm{g}\) of glucose contains about \(287\ \textrm{kJ}\) of chemical energy that originally came from sunlight.
This kind of estimate helps explain why photosynthesis matters at planetary scale. Tiny packets of stored energy in countless leaves, algae cells, and cyanobacteria add up to the energy foundation of the biosphere.
Glucose is a central product, but plants do not stop there. They can connect glucose molecules to form starch for storage or cellulose for cell walls. They can also convert photosynthetic products into lipids, proteins, pigments, and many other compounds. In other words, photosynthesis does not just produce "food"; it supports the building of living matter itself.
Wood, cotton, fruits, seeds, and paper all trace back to photosynthesis. A massive tree trunk is built largely from carbon atoms that once floated in the air as \(\textrm{CO}_2\). That sounds almost impossible at first, but it is true. Much of a plant's solid mass comes from gases taken from the atmosphere and converted into organic matter.
Most of the dry mass of a plant does not come from soil. It comes mainly from carbon dioxide in the air, transformed by photosynthesis into organic molecules.
The energy stored in plant molecules then moves through ecosystems. Herbivores eat plants, predators eat herbivores, decomposers break down dead organisms, and at each step some chemical energy is transferred while some is released as heat. But the original large-scale source of that energy is usually sunlight captured by photosynthesis.
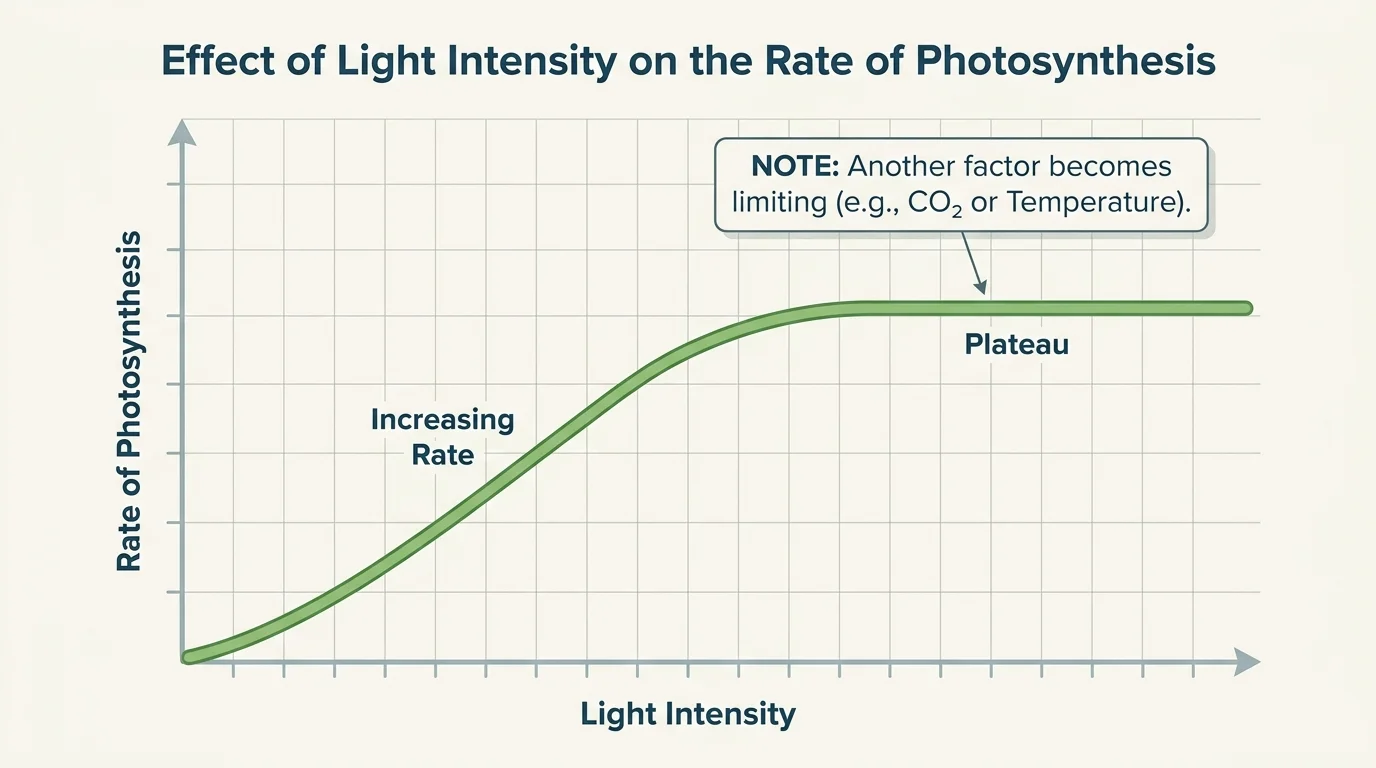
The rate of photosynthesis changes with environmental conditions, and [Figure 3] shows one common pattern: as a factor such as light intensity increases, the rate rises at first and then levels off when another factor becomes limiting.
Light intensity matters because photons drive the light-dependent reactions. At low light levels, increasing light usually increases the rate of photosynthesis. But beyond a certain point, more light may not help because chloroplasts are already working near capacity or because another factor, such as carbon dioxide availability, becomes the limit.
Carbon dioxide concentration also affects the rate because \(\textrm{CO}_2\) is a reactant in carbon fixation. If more carbon dioxide is available, the Calvin cycle can often proceed faster, up to a point. Temperature matters too because photosynthesis depends on enzymes, and enzyme activity changes with temperature.
Water availability is essential as well. When water is scarce, plants may close their stomata to reduce water loss. That also reduces carbon dioxide intake, which can slow photosynthesis. This is one reason drought can decrease plant growth even on bright sunny days.
The idea of a limiting factor is important. A process may depend on several requirements, but whichever requirement is in shortest supply relative to need sets the pace. The leveling-off pattern seen earlier in [Figure 3] is a classic sign that one variable is no longer the only control on the process.
| Factor | How it affects photosynthesis | Why |
|---|---|---|
| Light intensity | Usually increases rate at first | Provides energy for light reactions |
| Carbon dioxide concentration | Can increase rate up to a limit | Supplies carbon for sugar formation |
| Temperature | Has an optimal range | Enzymes work best within certain temperatures |
| Water availability | Low water often decreases rate | Water is a reactant and affects stomatal opening |
Table 1. Major factors that influence the rate of photosynthesis and the reason each matters.
Photosynthesis is not just a chapter in biology; it shapes agriculture, climate, and energy systems. Crop yields depend on how efficiently plants capture light, take in carbon dioxide, and convert those inputs into biomass. Farmers think about irrigation, nutrient supply, spacing, and greenhouse conditions partly because all of these influence photosynthetic performance.
Forests are enormous carbon reservoirs because photosynthesis removes carbon dioxide from the atmosphere and stores carbon in wood, roots, and soil. Oceans matter too. Microscopic photosynthetic organisms such as phytoplankton carry out a huge fraction of global photosynthesis. Even though they are tiny, their total effect is massive because there are so many of them.
Real-world case: a greenhouse
A greenhouse manager wants tomato plants to grow faster.
Step 1: Increase available light during short winter days with artificial lighting.
Step 2: Maintain a temperature range where photosynthetic enzymes work efficiently.
Step 3: Control water supply and sometimes raise carbon dioxide concentration inside the greenhouse.
Each action supports photosynthesis, which can increase sugar production and plant growth.
Biofuels are also connected to photosynthesis. Ethanol from corn and biodiesel from plant oils are forms of energy that ultimately come from recent biological capture of sunlight. Fossil fuels are different in timescale, but they too trace back to ancient photosynthesis. Coal, oil, and natural gas contain chemical energy originally stored by organisms millions of years ago.
This means that when a car burns gasoline, part of the released energy is sunlight from Earth's deep past. That idea is astonishing: modern machines can run on ancient photosynthetic energy compressed and transformed over geologic time.
Photosynthesis and cellular respiration are closely linked. Photosynthesis stores energy in glucose and other organic molecules. Cellular respiration releases some of that stored energy so cells can make ATP for immediate use.
A simplified equation for aerobic cellular respiration is:
\[\textrm{C}_6\textrm{H}_{12}\textrm{O}_6 + 6\textrm{O}_2 \rightarrow 6\textrm{CO}_2 + 6\textrm{H}_2\textrm{O} + \textrm{energy}\]
The two overall equations look almost opposite, but the actual processes are not simply reverse copies of each other. They use different enzymes, occur in different structures, and involve different pathways. Still, together they form a major cycle of matter and energy in living systems.
Plants carry out both photosynthesis and cellular respiration. During daylight, a plant may photosynthesize rapidly while also respiring continuously. At night, photosynthesis stops because there is no light, but cellular respiration continues.
One common misconception is that plants get their food from soil. Soil provides minerals and support, but the main carbon-based organic molecules are built inside the plant through photosynthesis. Another misconception is that oxygen is the main product. Oxygen is important, but for the plant the key result is the production of organic molecules that store energy and provide building material.
Another misunderstanding is that photosynthesis is only about plants. In reality, algae and photosynthetic bacteria are crucial, especially in aquatic systems. Much of the oxygen in Earth's atmosphere and much of global carbon fixation are linked to organisms many people never notice.
"Nearly all the energy for life comes from the Sun, but life depends on chemistry to hold that energy long enough to use it."
A simple observation can make the process more concrete. If an aquatic plant is placed in water under bright light, tiny bubbles may form on its leaves. Those bubbles are associated with oxygen being produced during photosynthesis. Changing the light level often changes how quickly the bubbles appear, linking visible evidence to the rate factors discussed earlier.
Photosynthesis is therefore more than a plant function. It is the main biological gateway through which solar energy enters the living world and becomes stored in matter. Without it, food webs, atmospheric oxygen at current levels, most ecosystems, and many human energy resources would be dramatically different or impossible.