Every bite of food, every sprint up a staircase, and every tree growing through concrete traces back to one of biology's most important ideas: life runs on energy transformations. Plants do not "make energy" from nothing, and your cells do not magically produce the power to think, move, repair damage, or maintain body temperature. Instead, living things capture, store, transfer, and release energy through linked chemical processes. The two central processes are photosynthesis and cellular respiration, and together they provide most of the energy used in life processes across ecosystems.
All organisms need energy to carry out life functions such as growth, transport, reproduction, movement, and maintaining internal balance. Cells use a molecule called ATP, or adenosine triphosphate, as an immediate energy source. ATP is often described as the cell's energy currency because it can be used quickly to power cellular work. But ATP is not stored in large amounts. Cells must constantly remake it.
Where does that energy ultimately come from? For most ecosystems on Earth, the original source is sunlight. Certain organisms capture light energy and store it in the chemical bonds of sugars. Then other organisms, including plants themselves, break down those sugars to release usable energy. That is why photosynthesis and cellular respiration are not separate topics. They are deeply connected parts of how ecosystems function.
Atoms are not created or destroyed in ordinary biological reactions. Matter is rearranged, while energy changes form. In living systems, carbon, hydrogen, and oxygen atoms are reorganized during photosynthesis and respiration, and energy is transferred through chemical bonds.
At the ecosystem level, this connection explains why producers are so important. They form the base of food webs because they convert energy from the environment into forms that other organisms can use. Consumers and decomposers depend on that stored chemical energy, directly or indirectly.
Photosynthesis is the process by which plants, algae, and some bacteria use light energy to build glucose from carbon dioxide and water. As [Figure 1] shows, this process depends on specialized structures inside cells that organize the steps of energy capture and sugar production. The overall equation is often written as:
\[6\textrm{CO}_2 + 6\textrm{H}_2\textrm{O} + \textrm{light energy} \rightarrow \textrm{C}_6\textrm{H}_{12}\textrm{O}_6 + 6\textrm{O}_2\]
This equation tells an important story. Carbon dioxide from the air and water from the environment are rearranged into glucose, a molecule that stores energy in its chemical bonds, and oxygen is released as a byproduct. The oxygen in Earth's atmosphere is largely a product of photosynthesis over billions of years.
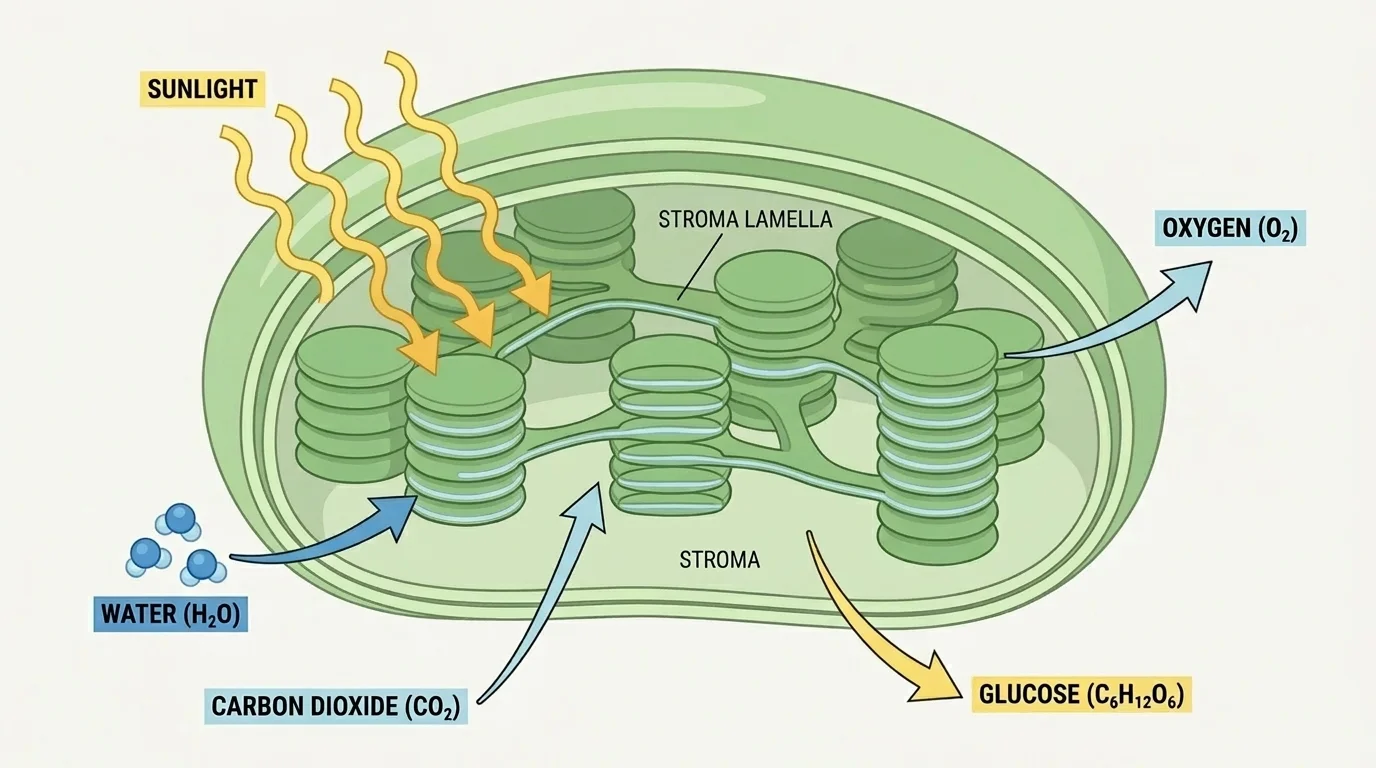
In plants, photosynthesis mainly occurs in leaves, especially in cells packed with chloroplasts. Chloroplasts contain chlorophyll, the green pigment that absorbs light most effectively in the red and blue parts of the visible spectrum and reflects green light. That reflected green light is why most leaves look green.
Producer means an organism that makes its own food using an energy source from the environment, usually sunlight. Pigment means a molecule that absorbs specific wavelengths of light. Glucose is a simple sugar that stores chemical energy and provides carbon skeletons for building other molecules.
Not all producers are large plants. Microscopic algae in oceans, lakes, and rivers perform an enormous share of global photosynthesis. In fact, much of the oxygen you breathe comes from photosynthetic organisms in water, not only from forests on land.
Photosynthesis can be understood in two linked sets of reactions. The first set is the light-dependent reactions, which occur in the thylakoid membranes of chloroplasts. These reactions absorb light energy and use it to split water molecules. This process releases electrons, hydrogen ions, and oxygen gas.
The light-dependent reactions produce energy-rich molecules that power the second set of reactions. A simplified way to think about it is that light energy is temporarily packaged into chemical forms the cell can use. Oxygen leaves the plant through tiny openings called stomata or dissolves into surrounding water if the producer is aquatic.
The second set is the Calvin cycle, which takes place in the stroma of the chloroplast. In this cycle, carbon dioxide is incorporated into organic molecules and eventually used to build glucose and other carbohydrates. Light is not directly required in every step of this cycle, but the cycle depends on products made by the light-dependent reactions.
Why glucose matters
Glucose is more than "plant food." It becomes fuel for respiration, raw material for cellulose in cell walls, starting material for starch storage, and part of many other biomolecules. Photosynthesis therefore supports both energy storage and the construction of living tissue.
A useful idea here is that photosynthesis stores energy, not just matter. The bonds formed in glucose contain energy originally captured from sunlight. When those bonds are later broken and reorganized during cellular respiration, the stored energy becomes available to cells.
For a simple numeric example, notice the coefficient pattern in the equation. If a plant uses \(6\) molecules of \(\textrm{CO}_2\), it forms \(1\) molecule of \(\textrm{C}_6\textrm{H}_{12}\textrm{O}_6\) and releases \(6\) molecules of \(\textrm{O}_2\). If the reactants double to \(12\textrm{CO}_2\) and \(12\textrm{H}_2\textrm{O}\), the products double to \(2\textrm{C}_6\textrm{H}_{12}\textrm{O}_6\) and \(12\textrm{O}_2\), assuming enough light is available.
Cellular respiration is the process by which cells break down glucose and transfer its stored energy into ATP. As [Figure 2] illustrates, the steps of aerobic respiration are divided among the cytoplasm and the mitochondria, allowing cells to harvest energy in a controlled way instead of all at once. The overall aerobic equation is essentially the reverse of photosynthesis:
\[\textrm{C}_6\textrm{H}_{12}\textrm{O}_6 + 6\textrm{O}_2 \rightarrow 6\textrm{CO}_2 + 6\textrm{H}_2\textrm{O} + \textrm{ATP energy}\]
Respiration happens in plants, animals, fungi, protists, and many bacteria. A common misunderstanding is that plants only photosynthesize while animals only respire. In reality, plants do both. They photosynthesize to make glucose, and they also respire to release energy from that glucose for their own cellular activities.
Cells do not usually use the energy in glucose directly for most work. Instead, they transfer it into ATP. When ATP loses one phosphate group, energy is released for activities such as muscle contraction, active transport across membranes, and synthesis of large molecules.
ATP is a molecule that stores usable energy for cells in phosphate bonds. Mitochondrion is an organelle where most aerobic respiration occurs in eukaryotic cells. Aerobic means requiring oxygen.
This staged release of energy is important. If all of glucose's energy were released at once, much would be lost as heat and could damage the cell. By breaking glucose down through many enzyme-controlled steps, cells capture more of that energy efficiently.
The first stage is glycolysis, which occurs in the cytoplasm. Glycolysis splits one glucose molecule into two smaller molecules called pyruvate. This stage does not require oxygen directly and produces a small amount of ATP.
The next major stage is the Krebs cycle, also called the citric acid cycle, which occurs in the mitochondrial matrix in eukaryotes. Before entering this cycle, pyruvate is further processed. The Krebs cycle releases carbon dioxide and transfers high-energy electrons to carrier molecules.
Those electrons move to the electron transport chain in the inner mitochondrial membrane. Oxygen is essential here because it acts as the final electron acceptor. This allows the chain to keep operating and drives the production of most ATP in aerobic respiration. Water forms when oxygen combines with electrons and hydrogen ions.
ATP yield example
The exact number varies by cell type and conditions, but many textbooks use about \(36\) to \(38\) ATP per glucose for aerobic respiration.
Step 1: Start with one glucose molecule.
One molecule of \(\textrm{C}_6\textrm{H}_{12}\textrm{O}_6\) enters glycolysis.
Step 2: Add the ATP contributions from stages.
Glycolysis makes a small amount, the Krebs cycle adds a little more, and the electron transport chain makes most of the ATP.
Step 3: State the approximate total.
A common estimate is about \(36\) ATP from one glucose molecule under aerobic conditions.
This shows why oxygen is so valuable for energy-demanding organisms: aerobic respiration captures far more usable energy than anaerobic pathways.
Here is a simple numerical comparison. If one glucose molecule yields about \(36\) ATP aerobically, and anaerobic fermentation yields only about \(2\) ATP, then aerobic respiration provides about \(18\) times as much ATP because \(\dfrac{36}{2} = 18\).
What happens when oxygen is unavailable or too limited? Cells can still get some energy through anaerobic processes, which do not require oxygen. In many organisms, the most important anaerobic pathway is fermentation.
Fermentation allows glycolysis to continue by regenerating molecules needed for that first stage. However, it does not produce large amounts of ATP. The ATP comes only from glycolysis, so the overall energy yield stays low.
In animal muscle cells, especially during intense exercise, lactic acid fermentation can occur when oxygen delivery cannot keep up with demand. Pyruvate is converted into lactic acid, allowing glycolysis to continue for a short time. This does not mean muscles "prefer" fermentation; it is more like an emergency backup system.
In yeast and some plant cells, alcoholic fermentation converts pyruvate into ethanol and carbon dioxide. This process is used in bread making and brewing. In bread dough, the \(\textrm{CO}_2\) bubbles make the dough rise.
Real-world fermentation examples
Step 1: Sprinting athlete
During a short, intense sprint, muscle cells may not receive oxygen fast enough. They continue making some ATP through glycolysis and lactic acid fermentation, which helps for brief bursts of activity.
Step 2: Bread dough
Yeast break down sugars anaerobically and release \(\textrm{CO}_2\). Gas bubbles expand the dough, while ethanol mostly evaporates during baking.
Step 3: Oxygen debt
After intense exercise, heavy breathing helps restore oxygen levels and supports the return to mainly aerobic respiration.
A quick numeric example makes the difference clear. If a cell needs about \(72\) ATP, it could get that from roughly \(2\) glucose molecules by aerobic respiration because \(2 \times 36 = 72\). Under fermentation, it would need about \(36\) glucose molecules because \(36 \times 2 = 72\). That huge difference explains why large, active organisms depend heavily on oxygen.
At the ecosystem scale, photosynthesis and respiration are partners. As [Figure 3] illustrates, photosynthesis removes carbon dioxide from the atmosphere or water and stores carbon in organic molecules, while respiration returns carbon dioxide to the environment. This continuous exchange is a major part of the carbon cycle.
Producers capture energy and store it in biomass. Consumers obtain that energy by eating producers or other consumers. Decomposers break down dead organisms and wastes, returning nutrients to ecosystems while also carrying out cellular respiration. Matter cycles, but energy flows through ecosystems and is eventually lost as heat.
This is why forests, grasslands, phytoplankton-rich oceans, and even microscopic communities in ponds matter so much. They are active sites of energy capture and carbon transformation. The same linked pattern seen in a chloroplast and a mitochondrion scales up to the biosphere.
Ocean phytoplankton are tiny, but collectively they carry out an enormous amount of global photosynthesis. Some estimates suggest they produce about half of Earth's oxygen.
The relationship is not a perfect mirror. Photosynthesis stores energy, while respiration releases it. Photosynthesis requires light, but respiration happens day and night. That is why plants still need oxygen and still respire in the dark, as the ecosystem cycle in [Figure 3] makes clear.
The rate of photosynthesis depends on several factors. Light intensity matters because light provides the energy for the reactions. At low light levels, increasing light often increases the rate. But after a certain point, another factor becomes limiting.
Carbon dioxide concentration also affects photosynthesis because carbon dioxide provides the carbon atoms used to build sugars. Temperature matters as well, because photosynthesis depends on enzymes, and enzyme activity changes with temperature. Too low and reactions slow down; too high and enzymes may lose function.
For respiration, the availability of oxygen strongly affects whether cells rely mainly on aerobic pathways or shift more toward anaerobic ones. Temperature also changes respiration rates in many organisms. For example, ectothermic animals such as insects and reptiles often show slower metabolism in cooler conditions and faster metabolism in warmer conditions, up to biological limits.
| Process | Main inputs | Main outputs | Where it occurs | Energy direction |
|---|---|---|---|---|
| Photosynthesis | \(\textrm{CO}_2\), \(\textrm{H}_2\textrm{O}\), light | \(\textrm{C}_6\textrm{H}_{12}\textrm{O}_6\), \(\textrm{O}_2\) | Chloroplasts of plants and algae; some bacteria use similar systems | Stores energy in glucose |
| Aerobic respiration | \(\textrm{C}_6\textrm{H}_{12}\textrm{O}_6\), \(\textrm{O}_2\) | \(\textrm{CO}_2\), \(\textrm{H}_2\textrm{O}\), ATP | Cytoplasm and mitochondria | Releases energy from glucose into ATP |
| Fermentation | Glucose breakdown products | Lactic acid or ethanol plus \(\textrm{CO}_2\) in some organisms, small ATP yield | Cytoplasm | Provides limited ATP without oxygen |
Scientists often investigate limiting factors with rate measurements. For example, if light intensity rises from \(20\) units to \(40\) units and oxygen production in a plant doubles from \(2\) to \(4\) bubbles per minute, light was likely limiting at first. If light rises again to \(80\) units but oxygen production stays near \(4\) bubbles per minute, some other factor, such as carbon dioxide concentration or temperature, has become limiting.
These processes shape real life in powerful ways. In agriculture, farmers use knowledge of photosynthesis to improve crop yields by managing light exposure, water supply, soil nutrients, and greenhouse carbon dioxide levels. In medicine and sports science, understanding respiration helps explain endurance, muscle fatigue, and recovery after intense exercise.
Biotechnology uses fermentation to produce bread, yogurt, cheese, biofuels, and some medicines. Environmental science relies on photosynthesis and respiration to understand climate change, carbon storage in forests, and oxygen levels in aquatic ecosystems. When excess nutrients cause algal blooms, respiration by decomposers can use up dissolved oxygen and create low-oxygen zones that harm fish and other organisms.
Why these processes matter for ecosystems
Photosynthesis brings usable energy into most ecosystems. Cellular respiration, including anaerobic pathways when necessary, makes that stored energy available to cells. Together they link individual survival to food webs, atmospheric chemistry, and global cycles of matter.
A forest, a loaf of bread, a cross-country race, and the oxygen in the atmosphere may seem unrelated at first. But all of them depend on the same chemistry of capturing and releasing energy.