A tree can build tons of wood mainly from carbon dioxide in the air. That sounds almost impossible until you realize that much of the solid material in a trunk began as invisible carbon dioxide in the atmosphere. Life on Earth depends on this kind of transformation: light becomes chemical energy, gases become sugars, and those sugars help build cells, tissues, and entire organisms. The same basic story connects green leaves, your muscles during a sprint, and the oxygen in the air you breathe.
All living things need both matter and energy. Matter provides the atoms used to build structures such as cell membranes, enzymes, DNA, and muscle fibers. Energy allows cells to do work: growing, repairing damage, moving materials, sending nerve signals, and maintaining internal balance. Organisms do not create matter or energy from nothing. Instead, they take in materials from their surroundings and transform energy from one form into another.
Plants, algae, and some bacteria are especially important because they can capture energy from sunlight and store it in chemical bonds. Animals, fungi, and many microorganisms then obtain that stored energy by eating food or breaking down organic compounds. In this way, life is powered by a continuous flow of energy and a constant recycling of atoms.
Photosynthesis is the process by which certain organisms use light energy to build sugars from carbon dioxide and water, releasing oxygen. Cellular respiration is the process by which cells break down food molecules, usually using oxygen, to transfer energy into ATP and release carbon dioxide and water. ATP is a molecule that stores usable energy for cell processes. The light-dependent reactions capture light energy, and the Calvin cycle uses that energy to help build sugars.
A useful way to think about this is that photosynthesis mostly stores energy, while cellular respiration mostly releases usable energy from that storage. These processes are not opposites in a simple mechanical sense, but they are deeply linked in the cycling of matter and the transfer of energy through living systems.
Chloroplasts are the organelles where most photosynthesis occurs in plants and many algae, and [Figure 1] shows how light, water, and carbon dioxide are brought into a system that produces sugar and oxygen. Chloroplasts contain chlorophyll and other pigments that absorb light energy, especially from the blue and red parts of the visible spectrum.
The overall chemical equation for photosynthesis is:
\[6\textrm{CO}_2 + 6\textrm{H}_2\textrm{O} + \textrm{light energy} \rightarrow \textrm{C}_6\textrm{H}_{12}\textrm{O}_6 + 6\textrm{O}_2\]
This equation shows that carbon dioxide and water are the reactants, and glucose and oxygen are the products. Glucose, written as \(\mathrm{C}_6\mathrm{H}_{12}\mathrm{O}_6\), is a sugar molecule that stores chemical energy in its bonds. The oxygen released during photosynthesis comes from water molecules that are split during the process.
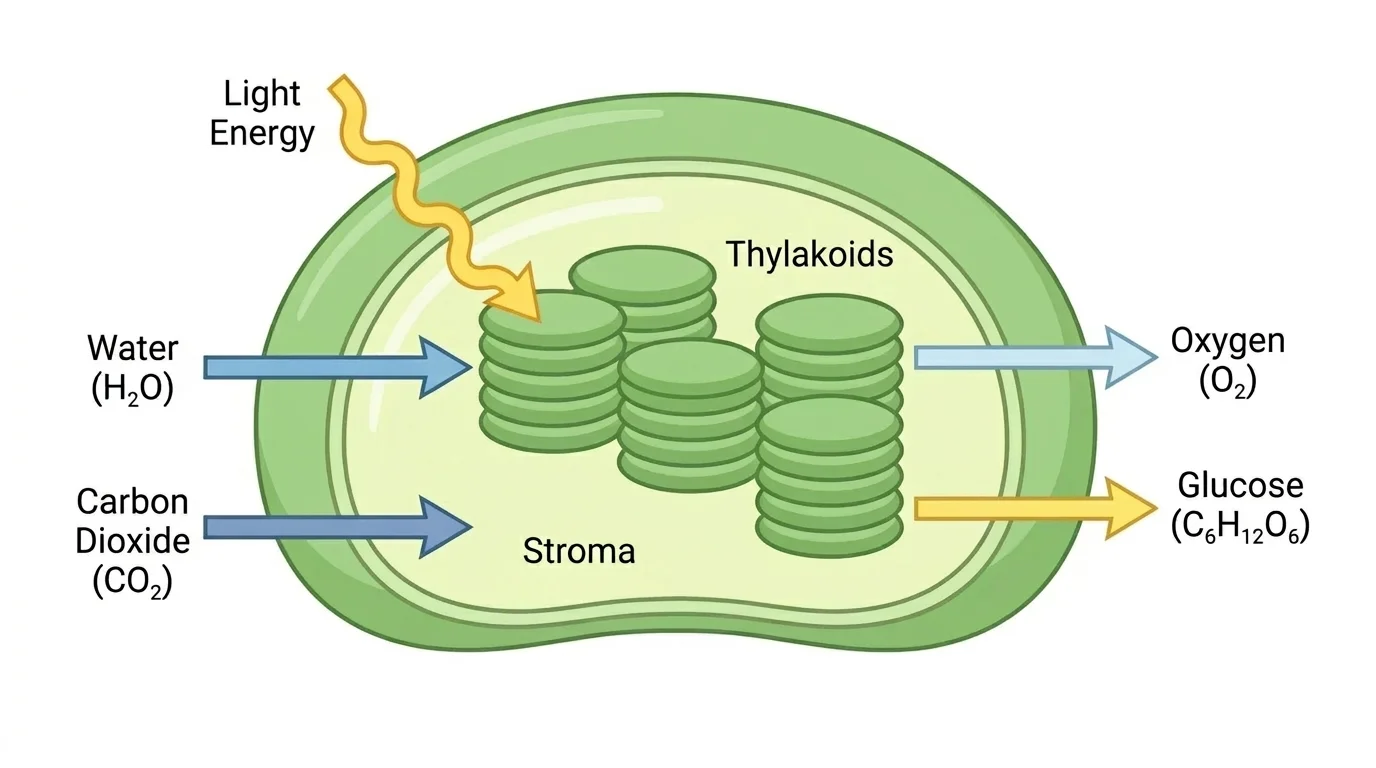
Photosynthesis does more than make food for a plant. It is the starting point for most food chains. A grass plant captures sunlight and makes sugars. A rabbit eats the grass. A fox eats the rabbit. At each step, energy originally captured from sunlight is passed through the ecosystem, though some of it is released as heat along the way.
Photosynthesis also changes Earth's atmosphere. Billions of years ago, photosynthetic organisms gradually increased the amount of oxygen in the air. That made aerobic respiration possible on a much larger scale, allowing organisms to release more usable energy from food.
Nearly all of the oxygen in Earth's atmosphere exists because photosynthetic organisms have been splitting water and releasing oxygen for immense spans of time. Much of that oxygen today is produced not by forests alone, but also by algae and photosynthetic microorganisms in oceans.
Even when people say plants "get food from the soil," that is only partly true. Soil provides minerals such as nitrogen, phosphorus, potassium, and iron, but most of a plant's mass comes from carbon fixed from atmospheric \(\mathrm{CO}_2\). The wood of a tree is therefore a record of carbon atoms pulled from air and assembled into new biological material.
Photosynthesis occurs in two connected stages, and [Figure 2] illustrates the flow between them. The first stage is the light-dependent reactions, which take place in the thylakoid membranes of the chloroplast. The second stage is the Calvin cycle, which occurs in the stroma, the fluid-filled area surrounding the thylakoids.
In the light-dependent reactions, pigments absorb light energy. That energy excites electrons and drives a series of reactions that split water molecules. When water is split, oxygen is released, and energy is transferred into molecules called ATP and NADPH. These molecules carry energy and high-energy electrons needed for the next stage.
In the Calvin cycle, carbon dioxide from the air is incorporated into organic molecules. Using the ATP and NADPH made earlier, the cycle builds higher-energy carbon compounds that can eventually be assembled into glucose and other sugars. So the light reactions capture energy, and the Calvin cycle uses that energy to build carbon-based molecules.
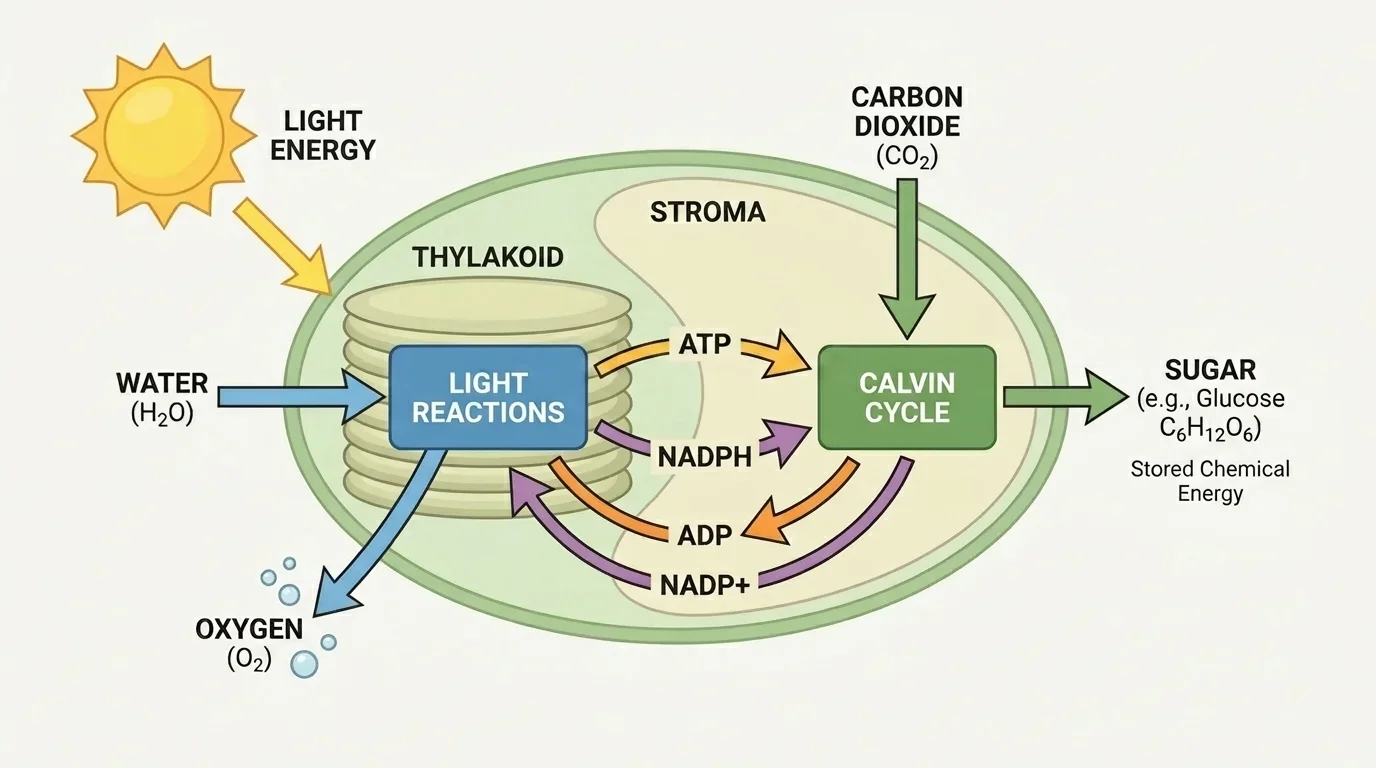
This process is a powerful example of energy conversion. Light energy does not become "food" directly. Instead, light energy drives electron movement, electron movement helps make ATP and NADPH, and those molecules provide the energy needed to form new chemical bonds in sugars. The stored energy in sugar can later be used by the plant itself or by another organism that eats the plant.
We can describe one simple numerical relationship using the photosynthesis equation. For every \(1\) glucose molecule formed, \(6\) molecules of \(\mathrm{CO}_2\) are required. If a plant cell makes \(2\) glucose molecules, it uses \(12\) molecules of \(\mathrm{CO}_2\) because \(2 \times 6 = 12\). This does not explain all the detailed intermediate steps, but it helps connect the balanced equation to real molecular counts.
Numeric example: using the photosynthesis equation
Suppose a leaf produces \(3\) glucose molecules. How many oxygen molecules are released according to the balanced equation?
Step 1: Use the ratio in the equation.
From \(6\mathrm{CO}_2 + 6\mathrm{H}_2\mathrm{O} \rightarrow \mathrm{C}_6\mathrm{H}_{12}\mathrm{O}_6 + 6\mathrm{O}_2\), each \(1\) glucose molecule corresponds to \(6\) oxygen molecules.
Step 2: Multiply by the number of glucose molecules.
\(3 \times 6 = 18\)
The leaf releases \(18\) molecules of \(\mathrm{O}_2\) in this simplified example.
The details of photosynthesis also explain why plants need both light and access to carbon dioxide and water. A plant in darkness cannot run the light reactions effectively. A plant with closed stomata on a hot, dry day may struggle to bring in enough \(\mathrm{CO}_2\). A wilted plant may also lack sufficient water for normal photosynthetic activity.
Glucose is not just a fuel. It is also a source of carbon atoms used to build many other molecules. The sugar molecules formed in photosynthesis contain carbon, hydrogen, and oxygen, arranged in a structure that can be modified into other carbon-based compounds. This is why the products of photosynthesis become the raw material for growth.
The carbon skeletons from sugars can be rearranged to form amino acids when nitrogen is added. Amino acids can then be linked together to make proteins. Proteins act as enzymes, structural fibers, transport molecules, and signaling molecules. In a growing plant, the carbon from glucose may become part of a new leaf cell wall, a chlorophyll-associated protein, or an enzyme in root tissue.
Sugars can also contribute to the formation of lipids and nucleotides. Lipids are important in membranes and energy storage. Nucleotides are the building blocks of DNA and RNA. This means the carbon fixed during photosynthesis can eventually become part of genes, membranes, and entire new cells.
As seen earlier in [Figure 1], the chloroplast produces sugar that leaves the organelle and enters the cell's larger economy of matter. A molecule built in one part of the cell may later become part of a tissue, an organ, or a seed. This is one of the key ideas in biology: atoms are reused in different combinations to create dramatically different structures.
| Starting material | Possible transformation | Biological result |
|---|---|---|
| Glucose | Stored as starch or moved as sucrose | Energy reserve or transport in plants |
| Carbon skeleton from sugar | Nitrogen added to form amino acids | Protein synthesis |
| Carbon from sugar | Rearranged into lipids | Cell membranes and long-term energy storage |
| Carbon from sugar | Used in nucleotide synthesis | DNA and RNA formation |
Table 1. Examples of how carbon from photosynthetic sugar can be incorporated into larger biological molecules.
Carbon backbones and biological diversity
Carbon is especially useful in living systems because it can form stable chains and rings. Those carbon backbones allow cells to build a huge variety of molecules with different shapes and functions. By adding different atoms or functional groups to carbon skeletons, organisms can create sugars, fats, proteins, pigments, hormones, and nucleic acids.
In animals, the same principle continues. When you eat a piece of fruit or bread, some of the molecules you absorb can be broken down for energy, while others provide building material. The carbon from food may later become part of your muscle proteins, cell membranes, or DNA during cell growth and repair.
Cellular respiration is the major process by which cells release usable energy from food molecules, and [Figure 3] shows how mitochondria transform the chemical energy in glucose into ATP while also releasing carbon dioxide, water, and heat. In eukaryotic cells, much of this process occurs in the mitochondria, the organelles primarily responsible for ATP production, though cells use ATP in many complex ways.
The overall equation for aerobic cellular respiration is:
\[\textrm{C}_6\textrm{H}_{12}\textrm{O}_6 + 6\textrm{O}_2 \rightarrow 6\textrm{CO}_2 + 6\textrm{H}_2\textrm{O} + \textrm{energy}\]
This equation does not mean cells simply burn glucose like a flame. Instead, respiration occurs through many enzyme-controlled steps. Bonds in glucose and intermediate compounds are broken, atoms are rearranged, and new compounds form. During this process, energy is transferred into ATP, which can power muscle contraction, active transport across membranes, synthesis of new molecules, and many other tasks.
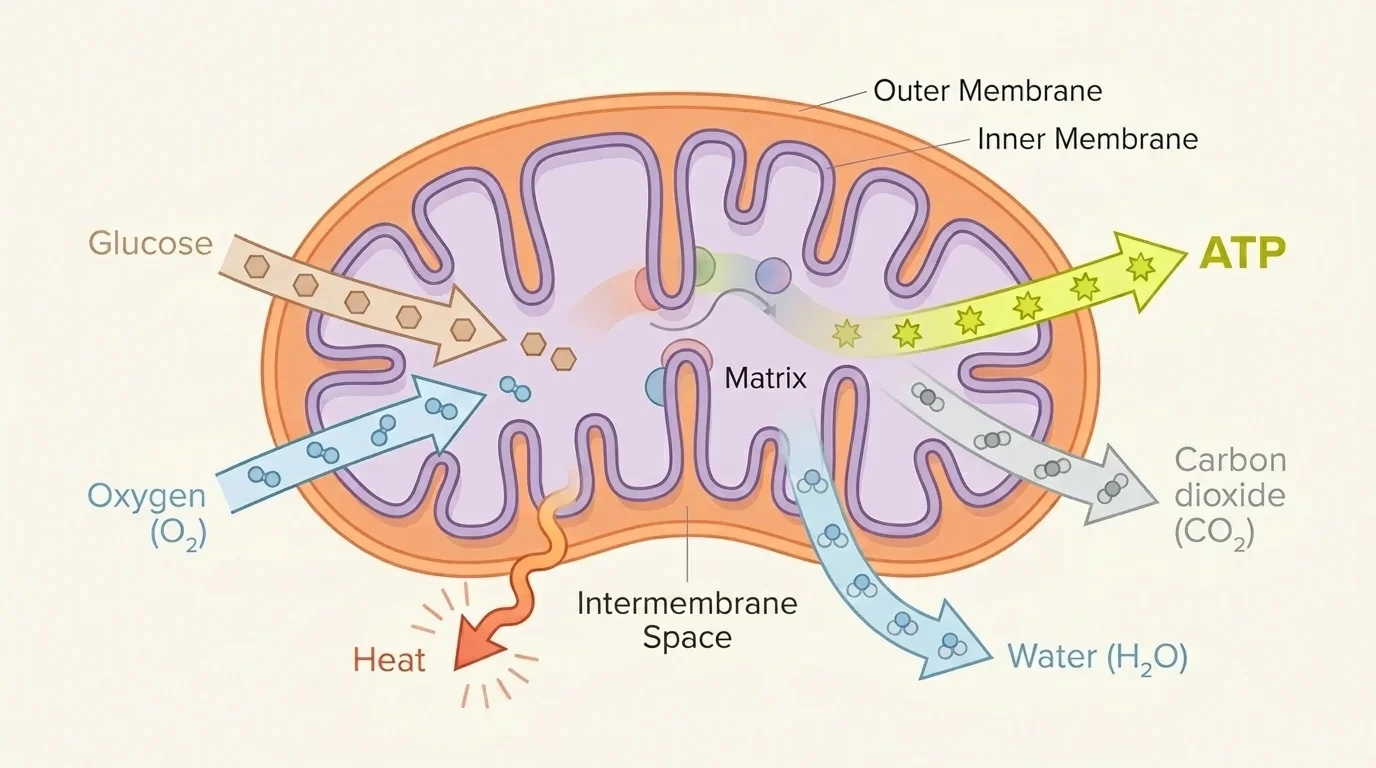
Aerobic respiration includes glycolysis, the citric acid cycle, and the electron transport chain. Glycolysis begins in the cytoplasm and splits glucose into smaller molecules. The citric acid cycle continues the breakdown in the mitochondrion. The electron transport chain uses high-energy electrons to create a gradient that drives ATP production. Oxygen is essential at the end of this chain because it accepts electrons and helps form water.
If oxygen is limited, some cells can use anaerobic pathways. Human muscle cells, for example, can temporarily rely more on fermentation during intense exercise. This provides less ATP than aerobic respiration and can contribute to muscle fatigue. The comparison helps explain why breathing rate increases during hard exercise: your body is trying to deliver enough oxygen to support rapid ATP production.
Numeric example: respiration ratios
If a cell completely breaks down \(4\) glucose molecules by aerobic respiration, how many molecules of \(\mathrm{CO}_2\) are produced according to the overall equation?
Step 1: Read the ratio from the equation.
Each \(1\) glucose molecule produces \(6\) molecules of \(\mathrm{CO}_2\).
Step 2: Multiply.
\(4 \times 6 = 24\)
The cell produces \(24\) molecules of \(\mathrm{CO}_2\) in this simplified calculation.
Respiration and photosynthesis are linked at the level of matter. Photosynthesis uses \(\mathrm{CO}_2\) and \(\mathrm{H}_2\mathrm{O}\) to build glucose and release \(\mathrm{O}_2\). Respiration uses glucose and \(\mathrm{O}_2\) to release energy and produce \(\mathrm{CO}_2\) and \(\mathrm{H}_2\mathrm{O}\). The atoms are not lost; they are rearranged into new combinations.
Not all the energy in food becomes useful cellular work. In every energy transfer, some energy is dispersed to the surroundings as heat. This is why your body warms up during exercise and why active organisms must manage heat gain and loss. The same principle applies in ecosystems: energy enters mainly as sunlight, is stored in chemical bonds, used by organisms, and gradually leaves as heat.
ATP is useful because it allows energy to be transferred in small, manageable amounts. When ATP is broken down to ADP and phosphate, cells can power specific reactions. But that transfer is never perfectly efficient. Some energy released from food molecules helps form ATP, and some is released as heat. In warm-blooded animals, this heat contributes to maintaining body temperature.
"Energy cannot be created or destroyed, only transformed."
— First Law of Thermodynamics
This principle helps explain why eating more food than the body needs does not make energy disappear. The chemical energy is transformed: some supports movement and metabolism, some is stored in molecules such as fat, and some is released as heat. During exercise, the demand for ATP rises sharply, so respiration speeds up and heat production increases.
For a simple numerical example, suppose a runner's cells use \(10\) units of chemical energy from food. If \(6\) units are transferred into ATP and cellular work, then \(4\) units are released as heat, since \(10 - 6 = 4\). Real cells are far more complex than this model, but the idea is essential: energy changes form, and some is always dispersed.
Biology makes the most sense when you track processes across scales, and [Figure 4] displays this movement from atoms to molecules to cells and larger structures. The carbon, hydrogen, oxygen, nitrogen, phosphorus, and sulfur in living things are organized into molecules; those molecules are organized into organelles and cells; cells build tissues; tissues form organs; and organs function within whole organisms.
When a plant grows a new leaf, atoms from water, carbon dioxide, and mineral nutrients are recombined into cellulose, proteins, chlorophyll, lipids, and DNA. Those molecules are assembled into cells, then into tissues such as epidermis, mesophyll, and vascular tissue. The same matter can continue moving through a food web if an insect eats the leaf and a bird eats the insect.
This means life is not made of special matter separate from the rest of the universe. Instead, ordinary chemical elements are repeatedly reorganized. Carbon atoms in a sugar molecule today may later be in a protein, then in a muscle cell, then returned to the atmosphere as \(\mathrm{CO}_2\) through respiration or decomposition.
The organization of matter also explains why changes at the molecular level can affect entire organisms. If a mutation alters the structure of a protein, a cell's function may change. If photosynthesis slows because drought limits \(\mathrm{CO}_2\) uptake, plant growth may decrease. If muscle cells cannot get enough oxygen, ATP supply drops and body performance suffers. Tiny molecular events can therefore scale up to visible biological outcomes.
Atoms are conserved in ordinary chemical reactions. The number of each kind of atom on one side of a balanced equation matches the number on the other side. What changes is how the atoms are bonded and arranged.
Seen this way, living systems are dynamic chemical systems. They constantly break bonds, form new ones, move matter between compartments, and transfer energy through reaction networks. The life of an organism depends on maintaining these flows in an organized way.
These ideas matter far beyond the biology classroom. In agriculture, farmers try to maximize photosynthesis by managing light exposure, water supply, soil nutrients, and carbon dioxide availability in some greenhouse settings. Higher photosynthetic rates can increase plant growth, but only if other limiting factors are also addressed.
In medicine and sports science, understanding respiration helps explain why trained athletes often have greater endurance. Their bodies can deliver oxygen more effectively, support aerobic respiration for longer periods, and manage ATP demands more efficiently. The burning sensation during intense effort is tied to metabolic conditions in muscle cells when oxygen delivery cannot fully match demand.
Climate science also depends on these processes. Forests, grasslands, wetlands, and oceans remove \(\mathrm{CO}_2\) from the atmosphere through photosynthesis. At the same time, organisms return \(\mathrm{CO}_2\) through respiration and decomposition. Human activities such as burning fossil fuels add large amounts of carbon dioxide, affecting the balance of the carbon cycle.
Aquatic ecosystems reveal another important connection. If algae photosynthesize rapidly in daylight, dissolved oxygen in water may rise. But if respiration by algae, bacteria, and other organisms becomes very high, especially at night, oxygen levels can fall. Fish and other aquatic animals may then experience stress or die if the water becomes too low in oxygen.
Case study: a seed becoming a seedling
Step 1: A seed begins with stored food molecules made earlier by the parent plant.
Step 2: During germination, the embryo uses cellular respiration to break down those stored molecules and make ATP.
Step 3: ATP powers growth, cell division, and the formation of new tissues.
Step 4: Once leaves develop, the young plant begins photosynthesis and starts producing more of its own sugars.
This sequence links stored matter, energy transfer, new cell formation, and the shift from dependence on stored food to direct use of sunlight.
Returning to [Figure 3], notice that respiration does not destroy matter or energy. It reorganizes matter and transfers energy into usable and less usable forms. Returning to [Figure 4], the same atoms can move across many organizational levels while still obeying the same chemical rules. That is one of biology's deepest themes: life is built from chemistry, but organized in ways complex enough to grow, respond, reproduce, and evolve.